1
answer
0
watching
638
views
12 Dec 2019
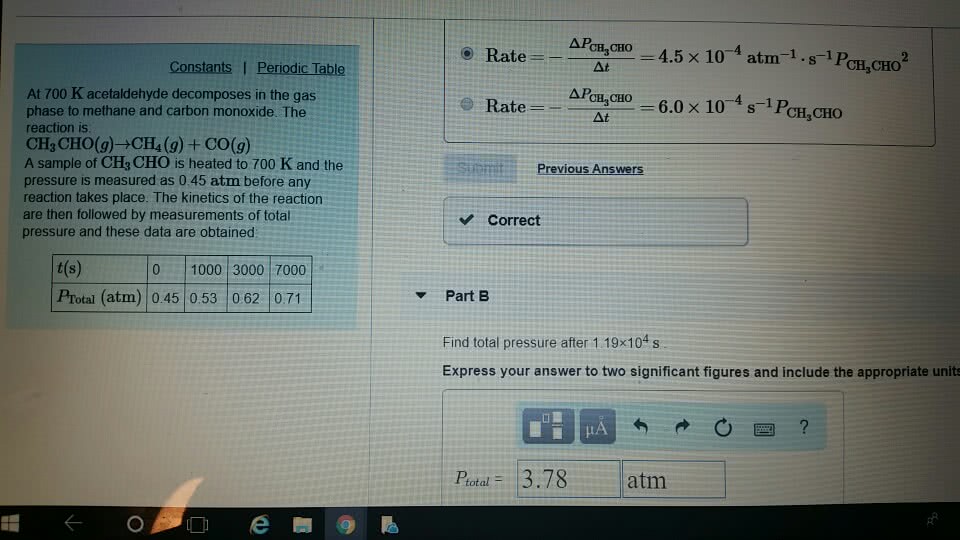
At 700 K acetaldehyde decomposes in the gas phase to methane and carbon monoxide. The reaction is: CH3CHO(g)âCH4(g)+CO(g) A sample of CH3CHO is heated to 700 K and the pressure is measured as 0.38 atm before any reaction takes place. The kinetics of the reaction are then followed by measurements of total pressure and these data are obtained:
t(s) 0 1000 3000 7000 PTotal (atm) 0.38 0.44 0.51 0.59
A. Find total pressure after 1.28Ã104s B. Find the rate law
At 700 K acetaldehyde decomposes in the gas phase to methane and carbon monoxide. The reaction is: CH3CHO(g)âCH4(g)+CO(g) A sample of CH3CHO is heated to 700 K and the pressure is measured as 0.38 atm before any reaction takes place. The kinetics of the reaction are then followed by measurements of total pressure and these data are obtained:
| t(s) | 0 | 1000 | 3000 | 7000 |
| PTotal (atm) | 0.38 | 0.44 | 0.51 | 0.59 |
A. Find total pressure after 1.28Ã104s B. Find the rate law
Reid WolffLv2
13 Dec 2019