3
answers
0
watching
1,929
views
23 May 2018
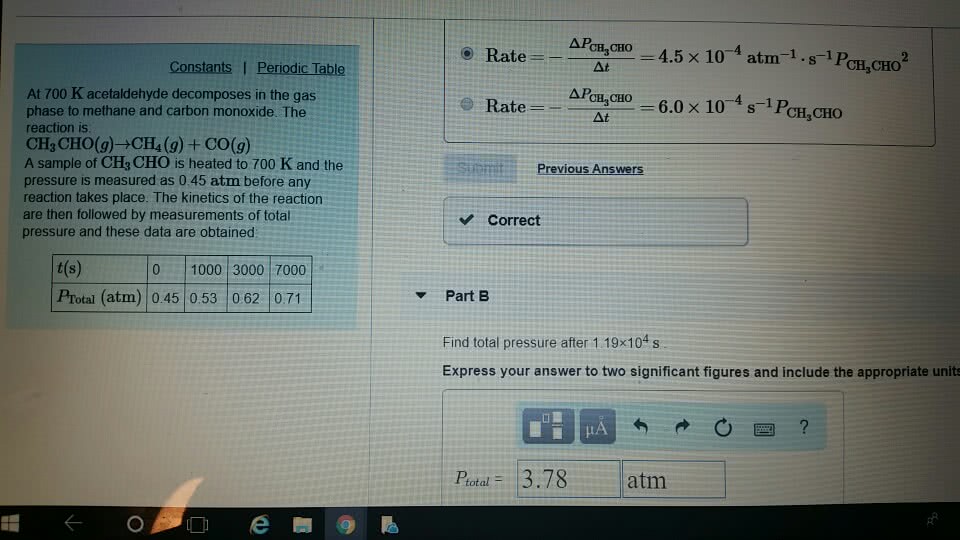
87. At 700 K acetaldehyde decomposes in the gas phase to methane and carbon monoxide. The reaction is: CH3CHO(g) → CH4(8) + CO(g) A sample of CH2CHO is heated to 700 K and the pressure is mea- sured as 0.22 atm before any reaction takes place. The kinetics of the reaction are followed by measurements of total pressure and these data are obtained: t(s) 0 1000 3000 7000 Protal (atm) 0.22 0.24 0.27 0.31 Find the rate law, the rate constant, and the total pressure after 2.00 x 10^s.
87. At 700 K acetaldehyde decomposes in the gas phase to methane and carbon monoxide. The reaction is: CH3CHO(g) → CH4(8) + CO(g) A sample of CH2CHO is heated to 700 K and the pressure is mea- sured as 0.22 atm before any reaction takes place. The kinetics of the reaction are followed by measurements of total pressure and these data are obtained: t(s) 0 1000 3000 7000 Protal (atm) 0.22 0.24 0.27 0.31 Find the rate law, the rate constant, and the total pressure after 2.00 x 10^s.
Read by 4 people
25 Jun 2023
Read by 6 people
Sixta KovacekLv2
26 May 2018
Already have an account? Log in