1
answer
0
watching
762
views
11 Dec 2019
Part 1:
a) write the ka reaction for HCNO.
b) write the equilibrium constant expression for the dissociation of HCNO.
c) Construct (don't solve) the ICE chart for the acid dissociation of 0.100 M HCNO.
d) what is the pH of a 0.100 M HCNO solution.
Part II:
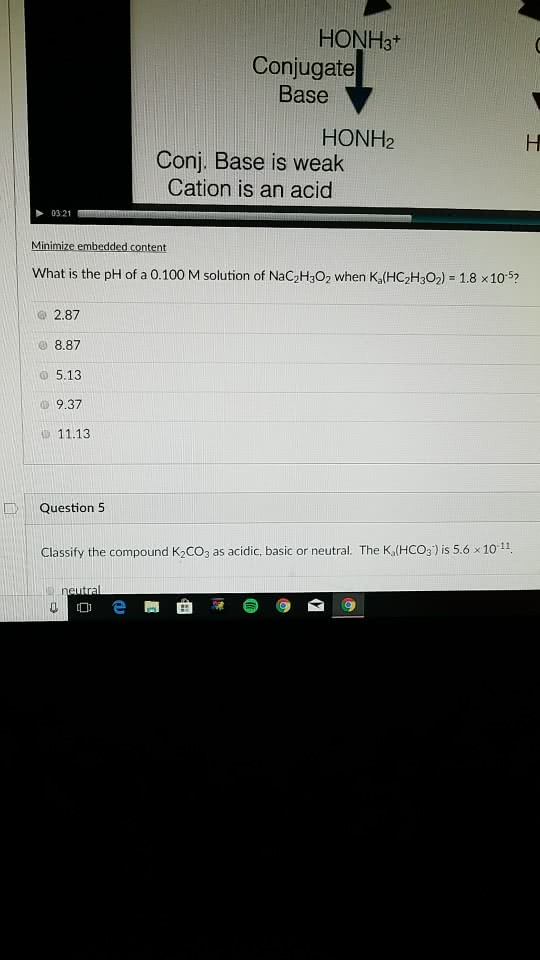
a) Write the base dissociation reaction of HONH2.
b) write the equilibrium constant expression for the base dissociation of HONH2.
c) Construct (don't solve) the ICE chart for the acid dissociation of 0.250 M HONH2.
d) what is the pH of 0.250 M HONH2.
Part 1:
a) write the ka reaction for HCNO.
b) write the equilibrium constant expression for the dissociation of HCNO.
c) Construct (don't solve) the ICE chart for the acid dissociation of 0.100 M HCNO.
d) what is the pH of a 0.100 M HCNO solution.
Part II:
a) Write the base dissociation reaction of HONH2.
b) write the equilibrium constant expression for the base dissociation of HONH2.
c) Construct (don't solve) the ICE chart for the acid dissociation of 0.250 M HONH2.
d) what is the pH of 0.250 M HONH2.
Trinidad TremblayLv2
13 Dec 2019