1
answer
2
watching
3,301
views
11 Nov 2019
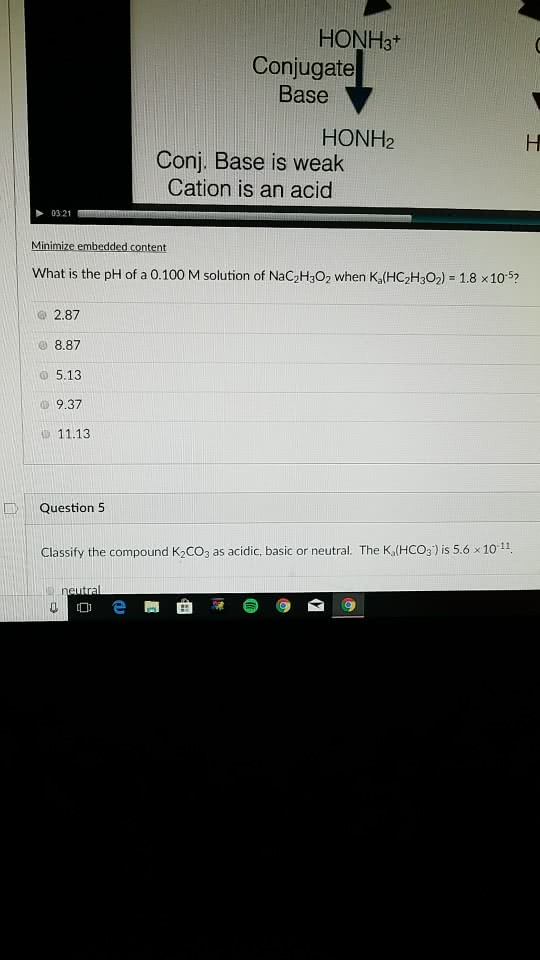
HONH3+ Conjugate Base HONH2 Conj. Base is weak Cation is an acid 03:21 Minimize embedded content What is the pH of a 0.100 M solution of NaC2H302 when Ka(HC2H3O2) 18 x105? 2.87 8.87 o 5.13 â© 9.37 D 11.13 Question 5 Classify the compound K2COs as acidic, basic or neutral. The K,lHCOg)is 5.6 1011

HONH3+ Conjugate Base HONH2 Conj. Base is weak Cation is an acid 03:21 Minimize embedded content What is the pH of a 0.100 M solution of NaC2H302 when Ka(HC2H3O2) 18 x105? 2.87 8.87 o 5.13 â© 9.37 D 11.13 Question 5 Classify the compound K2COs as acidic, basic or neutral. The K,lHCOg)is 5.6 1011
Verified Answer
Collen VonLv2
25 May 2019
Joey Tang
Bachelor’s Degree in Chemistry from McMaster University3 Jun 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.
