3
answers
0
watching
181
views
11 Dec 2019
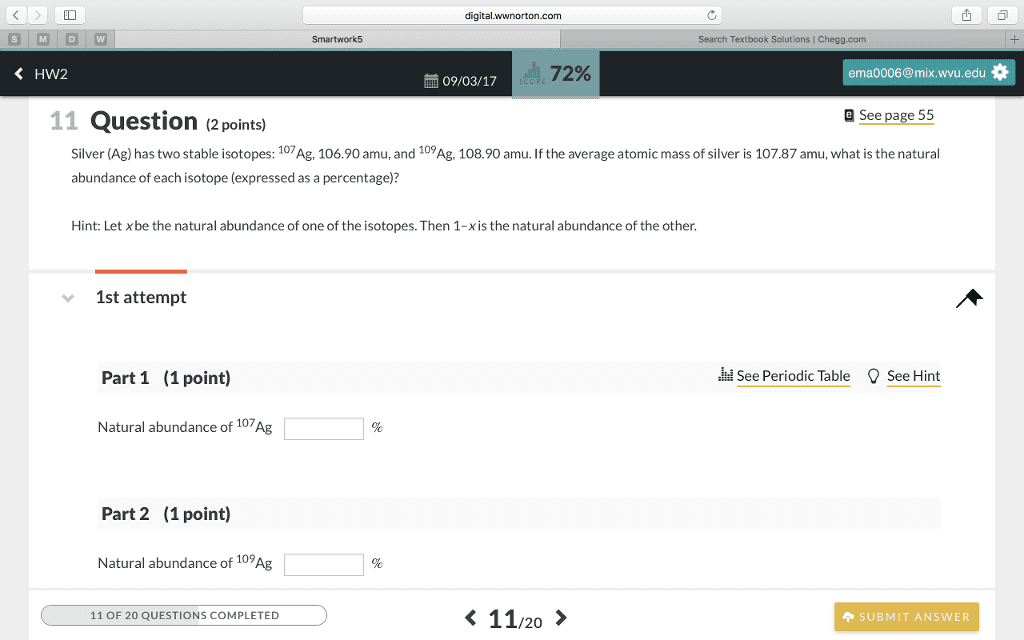
silver has two stable isotopes with the masses of 106.90509 and 108.9047. the atomic weight of silver is 107.868. what is the percent abundance of each isotope
silver has two stable isotopes with the masses of 106.90509 and 108.9047. the atomic weight of silver is 107.868. what is the percent abundance of each isotope
papayaprofessorLv10
5 Sep 2022
Irving HeathcoteLv2
13 Dec 2019
Already have an account? Log in