1
answer
0
watching
409
views
11 Dec 2019
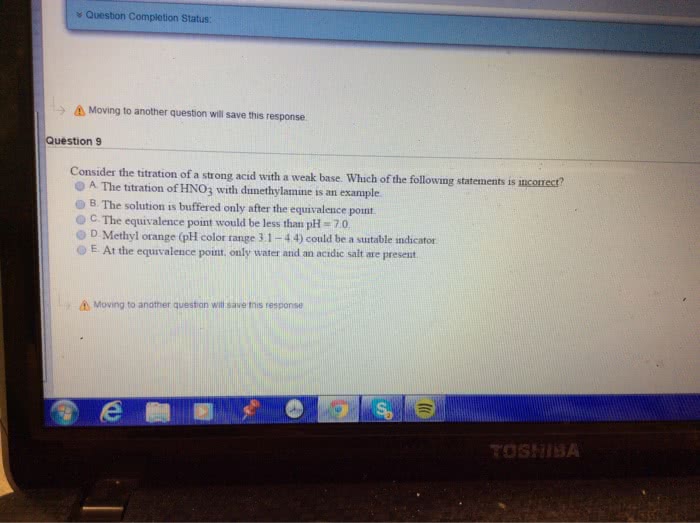
When titrating a monoprotic strong acid with a weak base at 25 degrees C, the
A) pH will be 7 at the equivalence point.
B) pH will be greater than 7 at the equivalence point.
C) titration will require more moles of the base than acid to reach the equivalence point.
D) titration will require more moles of acid than the base to reach the equivalence point.
E) pH will be less than 7 at the equivalence point.
When titrating a monoprotic strong acid with a weak base at 25 degrees C, the
A) pH will be 7 at the equivalence point.
B) pH will be greater than 7 at the equivalence point.
C) titration will require more moles of the base than acid to reach the equivalence point.
D) titration will require more moles of acid than the base to reach the equivalence point.
E) pH will be less than 7 at the equivalence point.
John Edward CayasLv10
30 Oct 2020