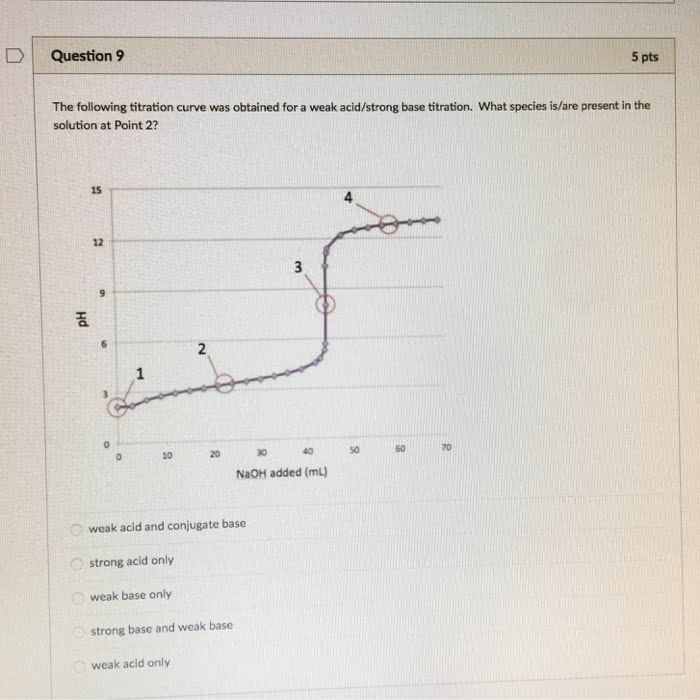
1
answer
0
watching
1,744
views
12 Dec 2019
Fill in the blanks with these words: base, H+, OH-, majority, minority, lower, greater, acid
The conjugate ______ of the weak acid is the ______ species in solution at the equivalence point, and this conjugate ______ reacts with water to produce _______ . Therefore, the pH at the equivalence point for a weak acid/strong base titration is ______ than 7.00.
Fill in the blanks with these words: base, H+, OH-, majority, minority, lower, greater, acid
The conjugate ______ of the weak acid is the ______ species in solution at the equivalence point, and this conjugate ______ reacts with water to produce _______ . Therefore, the pH at the equivalence point for a weak acid/strong base titration is ______ than 7.00.
Irving HeathcoteLv2
13 Dec 2019