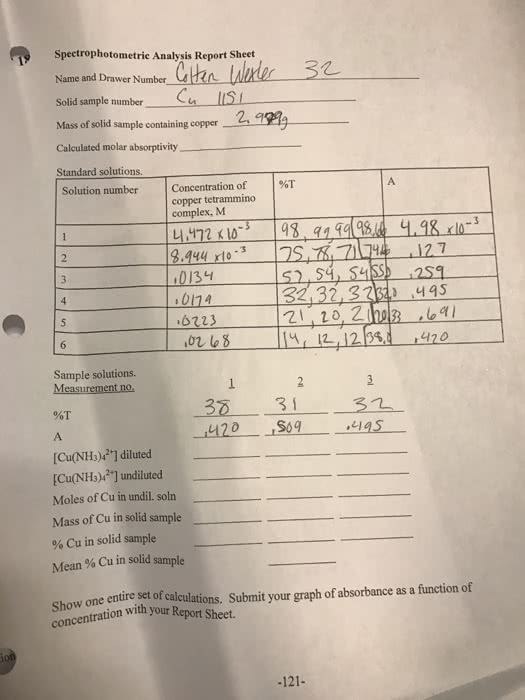
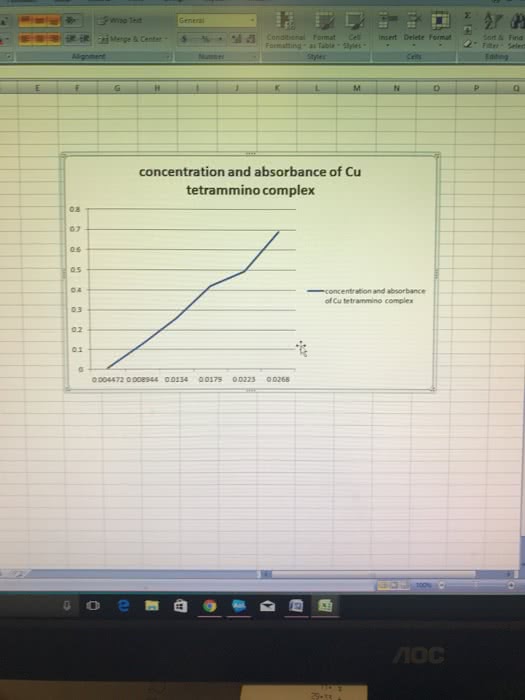
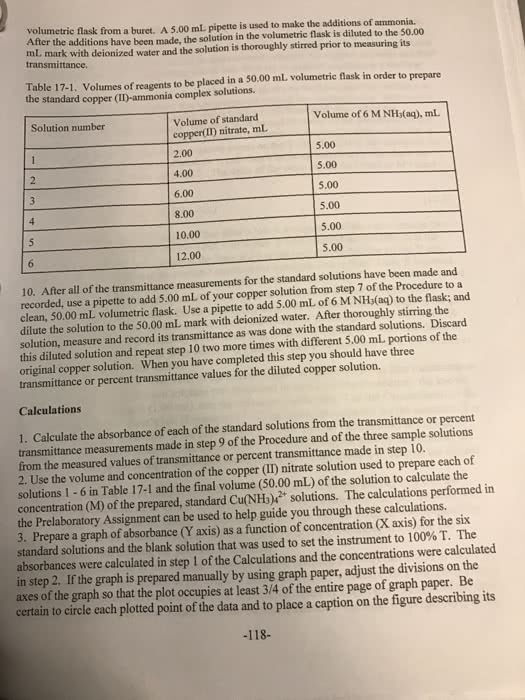
Beggining with the absorbance of your unkown brass sample show ur calculations for a) Cu+(2) (aq) concentration; b) mass of Cu in brass; c) percent Cu in brass, d) the average percent Cu in brass. Include caluclations for each trial and average when mass of brass is .725g and absorbance is .456 (nm) for first trial and for second trial mass of brass is .725g and absorbance is .525(nm)
Beggining with the absorbance of your unkown brass sample show ur calculations for a) Cu+(2) (aq) concentration; b) mass of Cu in brass; c) percent Cu in brass, d) the average percent Cu in brass. Include caluclations for each trial and average when mass of brass is .725g and absorbance is .456 (nm) for first trial and for second trial mass of brass is .725g and absorbance is .525(nm)
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
The lead (Pb) content in the sample is less than 25, and the Atomic Absorption Spectroscopy (AAS) method is applied to determine its content. One method is used to determine the Pb concentration in the given brass sample. In order to determine the lead content in the sample, 1 g brass sample was digested in concentrated nitric acid and diluted to 100 ml. 25.0 ml of this solution was diluted to 250.0 ml. 25.0 ml diluted sample solution was pipetted into five 50-ml volumetric flasks. Then, 0, 5, 10, 15 and 20 ml of 50 ppm Pb standard solution were added to each of the 50.0 ml volumetric flasks containing the sample solution. Dilute all the five flasks to the graduation mark with deionized water. The following table is the experimental results by AAS.
| Solution No. | Vol. of diluted sample solution, ml | Vol. of 50 ppm Pb, ml | Conc. of Pb, ppm | Absorbance |
| 1 | 25.0 | 0 | 0.080 | |
| 2 | 25.0 | 5.0 | 0.120 | |
| 3 | 25.0 | 10.0 | 0.178 | |
| 4 | 25.0 | 15.0 | 0.240 | |
| 5 | 25.0 | 20.0 | 0.310 |
Show all of your calculation steps.
(a) (i) Find out the concentration of Pb (in ppm) for each of the solutions stated in the table above.
(ii) Draw a graph, and determine the unknown concentration from the graph. Then, calculate the %Pb present in the brass sample.
(b) Explain why the sample solution is not prepared separately, buy together with standard lead solution? Give TWO reasons to explain why the results obtained under this method are better than using external standard method.
Just need help answering the ***** question under conclusions. here is my worksheet with information
Record your absorbance measurements for the copper (II) sulfate solution in the table below.
| Wavelength (nm) | Absorbance |
| 400 | 0.00 nm |
| 420 | 0.00 nm |
| 440 | 0.00 nm |
| 460 | 0.00 nm |
| 480 | 0.00 nm |
| 500 | 0.00 nm |
| 520 | 0.00 nm |
| 540 | 0.00 nm |
| 560 | 0.02 nm |
| 580 | 0.05 nm |
| 600 | 0.09 nm |
| 620 | 0.14 nm |
| 640 | 0.22 nm |
| 660 | 0.35 nm |
| 680 | 0.50 nm |
| 700 | 0.63 nm |
Record your absorbance measurements for the cobalt (II) chloride solution in the table below.
| Wavelength (nm) | Absorbance |
| 400 | 0.00 nm |
| 420 | 0.1 nm |
| 440 | 0.07 nm |
| 460 | 0.07 nm |
| 480 | 0.16 nm |
| 500 | 0.23 nm |
| 520 | 0.22 nm |
| 540 | 0.13 nm |
| 560 | 0.07 nm |
| 580 | 0.04 nm |
| 600 | 0.00 nm |
| 620 | 0.00 nm |
| 640 | 0.00 nm |
| 660 | 0.00 nm |
| 680 | 0.00 nm |
| 700 | 0.00 nm |
Data Analysis
Create and save a graph of absorbance (y-axis) versus the wavelength (x-axis) for both copper (II) sulfate and cobalt (II) chloride. Click the graphing icon below to create your graph. 
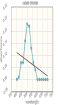
Experiment 2: Measure Absorbance versus Concentration for Cu+2 Ions
Lab Results
What wavelength did you use to measure the absorbance of the copper (II) sulfate solutions?
| 700 nm |
Data Analysis
Create and save a graph of absorbance (y-axis) versus the concentration of the copper (II) sulfate solutions (x-axis). Click the graphing icon below to create your graph. 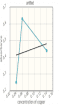
The y-intercept of the plot of absorbance versus concentration is approximately 0. Why is this?
| It should be a straight line obtained by having a slope of = 1 and y-intercept = 0. |
Experiment 3: Determine the Cu+2 Concentration in Unknown Solutions of Copper (II) Sulfate
Lab Results
How many mL of the unknown solutions did you use for your absorbance measurements?
| 10 mL |
Data Analysis
Determine the copper (II) sulfate concentrations in your two unknown samples from the calibration plot.
| wavelength used: 700 nm absorbance unknown: a. 0.08 b. 0.12 |
Conclusions
*******Were the unknown solutions more or less concentrated than the copper (II) sulfate solution used in experiment 1?
Given the data in the table below, what is the concentration of the unknown solution? Both solutions contain the same substance.
| standard solution concentration | 1.25 M |
| standard solution absorbance | 0.75 |
| unknown solution absorbance | 0.38 |
| 0.63 M is the concentration of the unknown solution. |