1
answer
0
watching
321
views
19 Nov 2019
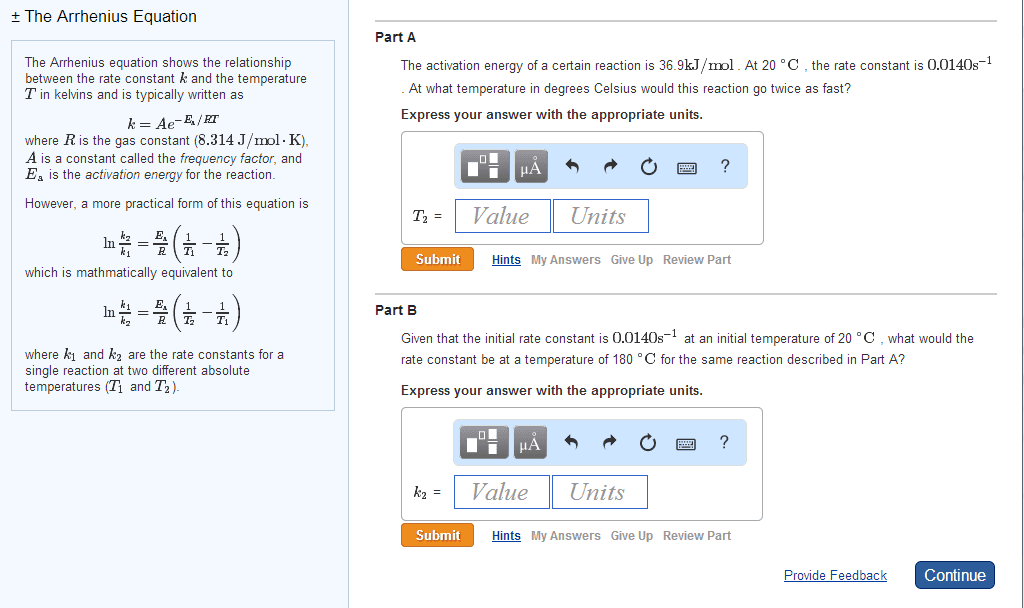
2. Using the Arrhenius equation, k = A e(^ -(Ea/RT)) a. Derive the equation of the plot of ln k vs. 1/T . b. Knowing that the value of the rate constant (k) for a certain reaction is 1.35ï´10ï4 sï1 at 35ï°C, and A= 2.6ï´1013 s-1, what is the activation energy? R= 8.314 J/K.mol c. Determine the half-life of the above reaction at the given temperature
2. Using the Arrhenius equation, k = A e(^ -(Ea/RT)) a. Derive the equation of the plot of ln k vs. 1/T . b. Knowing that the value of the rate constant (k) for a certain reaction is 1.35ï´10ï4 sï1 at 35ï°C, and A= 2.6ï´1013 s-1, what is the activation energy? R= 8.314 J/K.mol c. Determine the half-life of the above reaction at the given temperature
Lelia LubowitzLv2
8 Jun 2019