1
answer
0
watching
129
views
17 Nov 2019
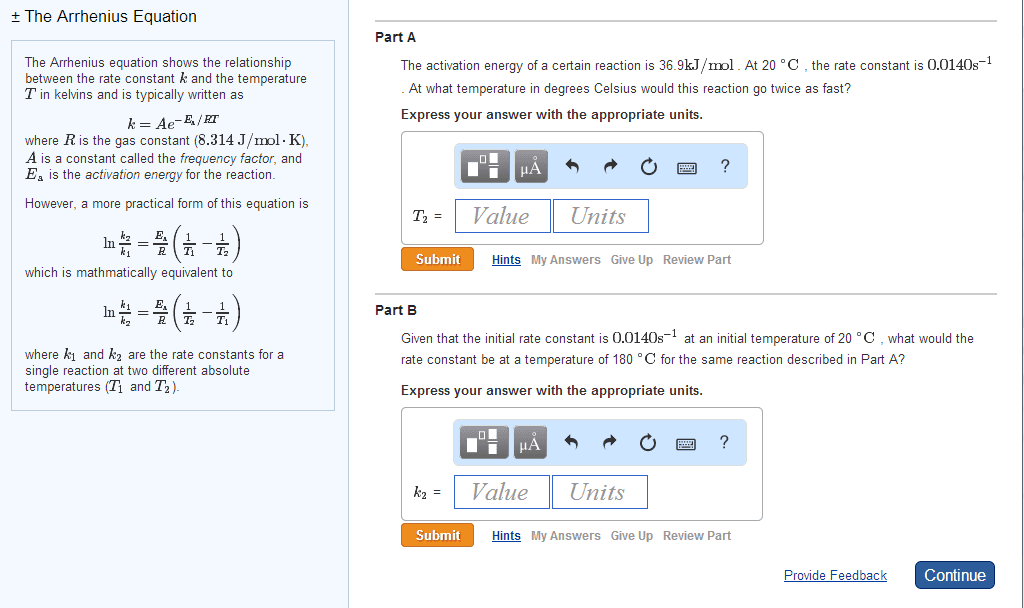
Use the following equation and chart to make a graph and obtain Ea in kJ/mol for this data. [My main problem is setting up and solving the equation mostly because I don't know where A is comming from and I don't know what to put.]
ln(k)=(-Ea/R)(1/T)+ln(A)
k=rate constant
Ea=activation energy
R=gas constant, 8.314 J/(mol K)
T=temperature in Kelvin
A=Arrhenius constant
T(Kelvin) k1(sec-1) k2(sec-1) k average(sec-1) 300 71 67 69.0 320 112 99 105.5 290 59 58 58.5
Use the following equation and chart to make a graph and obtain Ea in kJ/mol for this data. [My main problem is setting up and solving the equation mostly because I don't know where A is comming from and I don't know what to put.]
ln(k)=(-Ea/R)(1/T)+ln(A)
k=rate constant
Ea=activation energy
R=gas constant, 8.314 J/(mol K)
T=temperature in Kelvin
A=Arrhenius constant
| T(Kelvin) | k1(sec-1) | k2(sec-1) | k average(sec-1) |
|---|---|---|---|
| 300 | 71 | 67 | 69.0 |
| 320 | 112 | 99 | 105.5 |
| 290 | 59 | 58 | 58.5 |
Bunny GreenfelderLv2
17 Jul 2019