3
answers
0
watching
92
views
28 Sep 2019
This question is also already on the cramster site, but I don'tunderstand the answer.
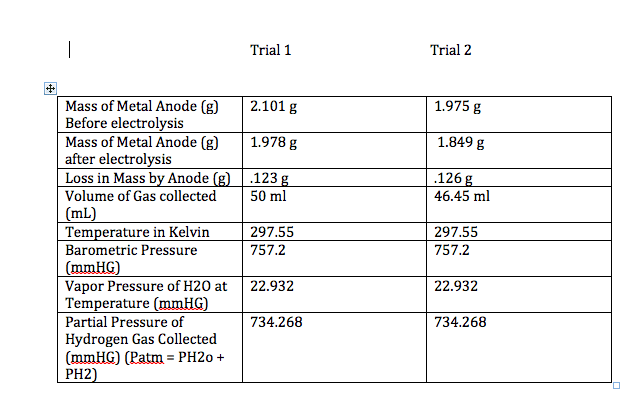
Ch. 17 #73: What is the reduction potential at 25 C for thehydrogen electrode in each of the following solutions? The halfreaction is:
2H+ (aq) + 2 e- --> H2 (g, 1 atm)
E = E^ - 0.0592V/n x log pH2/[H+]^2 b.) a solution having pH = 4.00 Please explain why the number of moles of electrons (n) is 1and why in the worked answer, the concentration of hydrogen [10^-4]is not squared as the hydrogen concentration was for the halfreaction given. i.e. answer: E = E^ - 0.0592V/1 x log (1/[10^-4} So in short, why is n = 1 moles of electrons and why is it log(1/[10^-4]) and not log (1/[10^-4}^2)? Thanks.
This question is also already on the cramster site, but I don'tunderstand the answer.
Ch. 17 #73: What is the reduction potential at 25 C for thehydrogen electrode in each of the following solutions? The halfreaction is:
2H+ (aq) + 2 e- --> H2 (g, 1 atm)
Ch. 17 #73: What is the reduction potential at 25 C for thehydrogen electrode in each of the following solutions? The halfreaction is:
2H+ (aq) + 2 e- --> H2 (g, 1 atm)
E = E^ - 0.0592V/n x log pH2/[H+]^2
b.) a solution having pH = 4.00
Please explain why the number of moles of electrons (n) is 1and why in the worked answer, the concentration of hydrogen [10^-4]is not squared as the hydrogen concentration was for the halfreaction given.
i.e. answer: E = E^ - 0.0592V/1 x log (1/[10^-4}
So in short, why is n = 1 moles of electrons and why is it log(1/[10^-4]) and not log (1/[10^-4}^2)?
Thanks.
3
answers
0
watching
92
views
For unlimited access to Homework Help, a Homework+ subscription is required.
bhavish0602Lv10
26 Apr 2023
25 Apr 2023
Already have an account? Log in
Trinidad TremblayLv2
28 Sep 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232