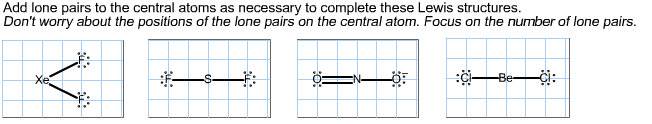
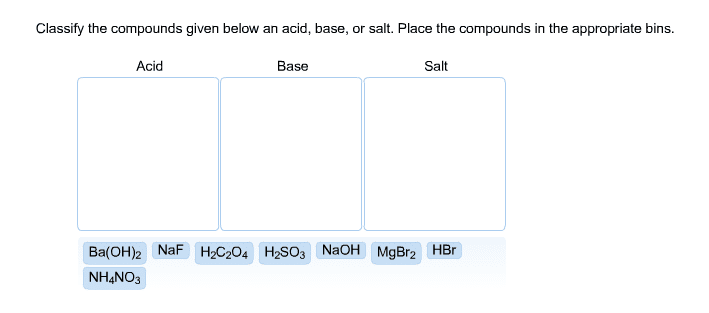
2
answers
0
watching
1,032
views
6 Nov 2019
Draw Lewis structures of the four molecules and/or ions with theformulas given below and figure out which two of these areisoelectronic with each another. Species which are isoelectronichave the same number of atoms, the same total number of electrons,the same number of valence electrons, and the same valence electronstructure (single and multiple bonds and lone pairs in sameplaces), but need not have the same distribution of formal chargeson atoms.
C22- and N2 and O2 and OCl-
a) N2 and OCl- are isoelectronic.
b) C22- and O2 are isoelectronic.
c) N2 and O2 are isoelectronic.
d) N2 and C22- are isoelectronic.
e) C22- and OCl- are isoelectronic.
Draw Lewis structures of the four molecules and/or ions with theformulas given below and figure out which two of these areisoelectronic with each another. Species which are isoelectronichave the same number of atoms, the same total number of electrons,the same number of valence electrons, and the same valence electronstructure (single and multiple bonds and lone pairs in sameplaces), but need not have the same distribution of formal chargeson atoms.
C22- and N2 and O2 and OCl-
a) N2 and OCl- are isoelectronic.
b) C22- and O2 are isoelectronic.
c) N2 and O2 are isoelectronic.
d) N2 and C22- are isoelectronic.
e) C22- and OCl- are isoelectronic.
C22- and N2 and O2 and OCl-
a) N2 and OCl- are isoelectronic.
b) C22- and O2 are isoelectronic.
c) N2 and O2 are isoelectronic.
d) N2 and C22- are isoelectronic.
e) C22- and OCl- are isoelectronic.
mahayodha80Lv10
1 Aug 2022
Collen VonLv2
23 Jul 2019
Already have an account? Log in