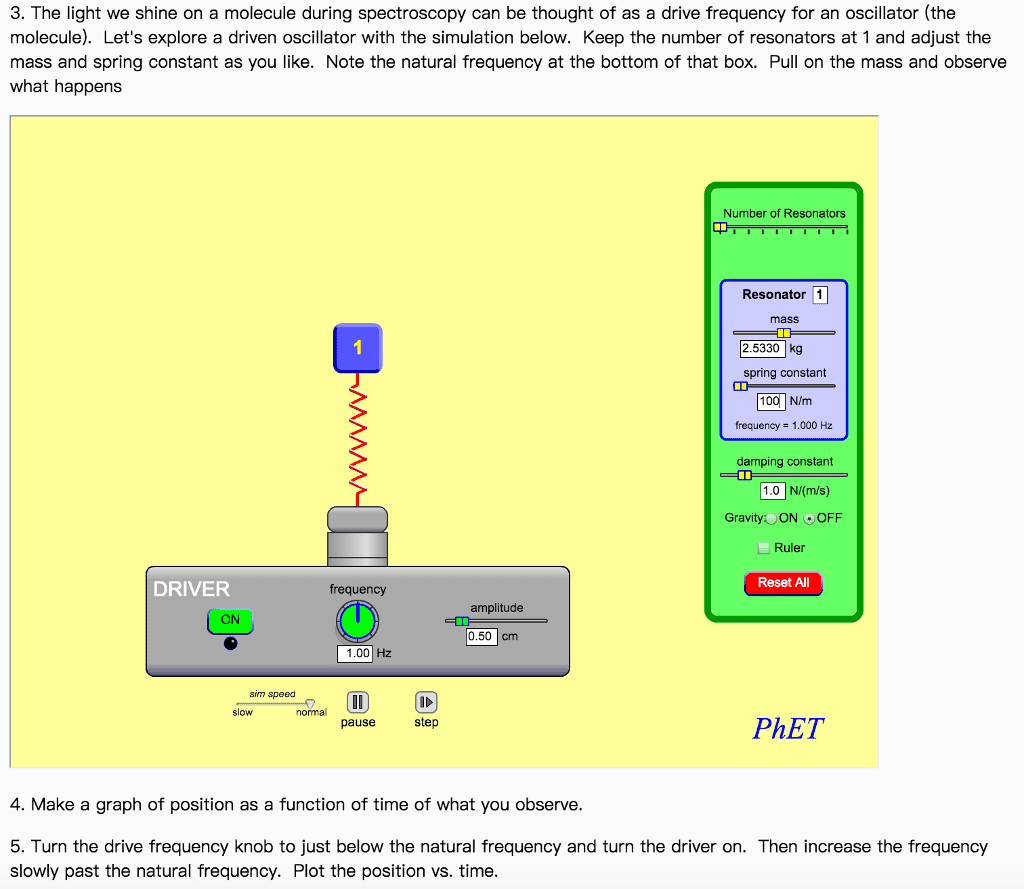
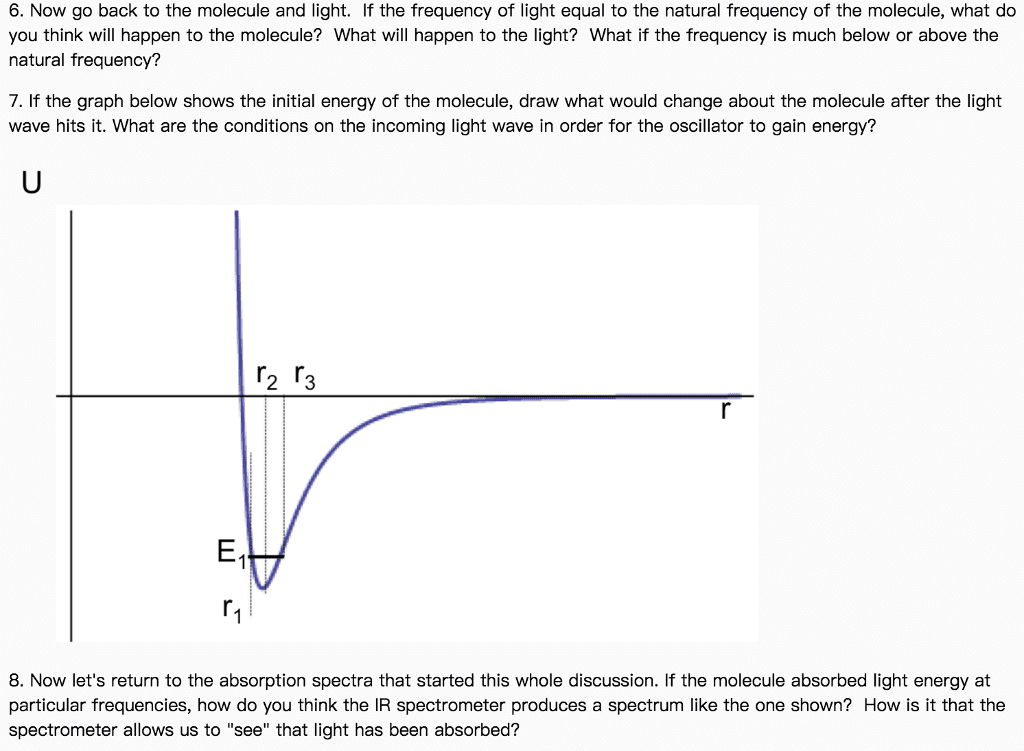
7
answers
0
watching
2,203
views
28 Oct 2017
73. Ultraviolet radiation and radiation of shorter wavelengths can damage biological molecules because these kinds of radiation carry enough energy to break bonds within the molecules. A typical carbon-carbon bond requires 348 kJ/mol to break. What is the longest wavelength of radiation with enough ener gy to break carbon-carbon bonds?
73. Ultraviolet radiation and radiation of shorter wavelengths can damage biological molecules because these kinds of radiation carry enough energy to break bonds within the molecules. A typical carbon-carbon bond requires 348 kJ/mol to break. What is the longest wavelength of radiation with enough ener gy to break carbon-carbon bonds?
jagdishkumarLv7
15 Jul 2023
26 Feb 2023
Already have an account? Log in
24 Feb 2023
Already have an account? Log in
Read by 1 person
Nelly StrackeLv2
30 Oct 2017
Already have an account? Log in