1
answer
0
watching
1,517
views
28 Sep 2019
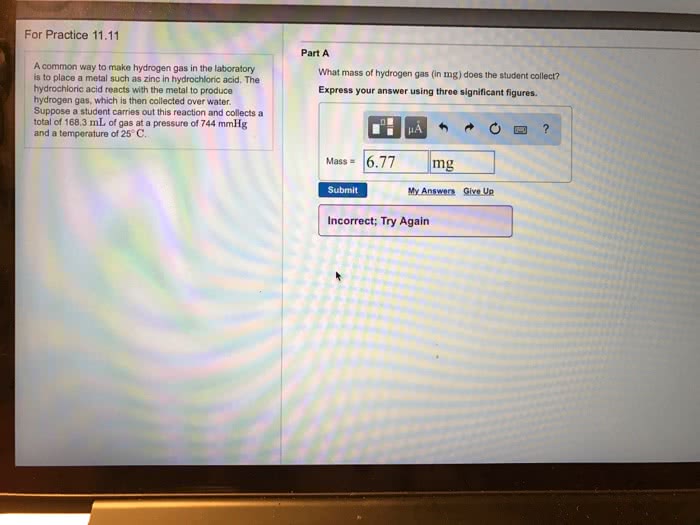
Modern pennies are composed of zinc and coated with copper. After making several scratches in a penny, a student determines the mass of a penny to be 2.482g. The student puts the penny into hydrochloric acid, where the zinc (but not the copper) reacts with hydrochloric acid via a single replacement reaction. The hydrogen is collected at 25 degrees Celsius over water. The collected gas occupies 0.899L at a total pressure of 791mm Hg. Calculate the percent zinc (by mass) in the penny. (Assume that all of the zinc in the penny dissolves.)
Modern pennies are composed of zinc and coated with copper. After making several scratches in a penny, a student determines the mass of a penny to be 2.482g. The student puts the penny into hydrochloric acid, where the zinc (but not the copper) reacts with hydrochloric acid via a single replacement reaction. The hydrogen is collected at 25 degrees Celsius over water. The collected gas occupies 0.899L at a total pressure of 791mm Hg. Calculate the percent zinc (by mass) in the penny. (Assume that all of the zinc in the penny dissolves.)
Verified Answer
Jean KeelingLv2
30 Sep 2019
23 Oct 2020
Answer verification
This is a step by step verification of the answer by our certified expert.
Subscribe to our livestream channel for more helpful videos.