101 Lecture Notes - Lecture 1: Magnetic Resonance Imaging, Louis De Broglie, Quantum Decoherence
Document Summary
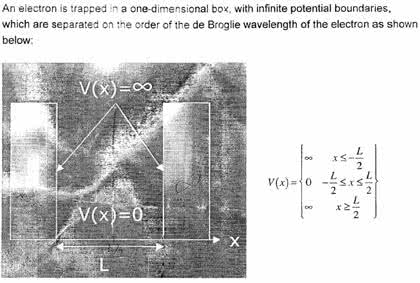
Quantum physics is a branch of physics that studies the behavior of matter and energy at the atomic and subatomic levels. It describes the strange and fascinating properties of the smallest particles in the universe, such as electrons, protons, and photons, which can exist in multiple states simultaneously and interact in ways that defy our classical understanding of physics. The foundation of quantum mechanics is based on the wave-particle duality of matter, which means that particles can exhibit wave-like properties and waves can exhibit particle-like properties. This idea was first introduced by the french physicist louis de broglie in 1924 and was later confirmed experimentally by the famous double-slit experiment. In quantum mechanics, the state of a particle is described by a wave function, which gives the probability of finding the particle in a particular location or state.