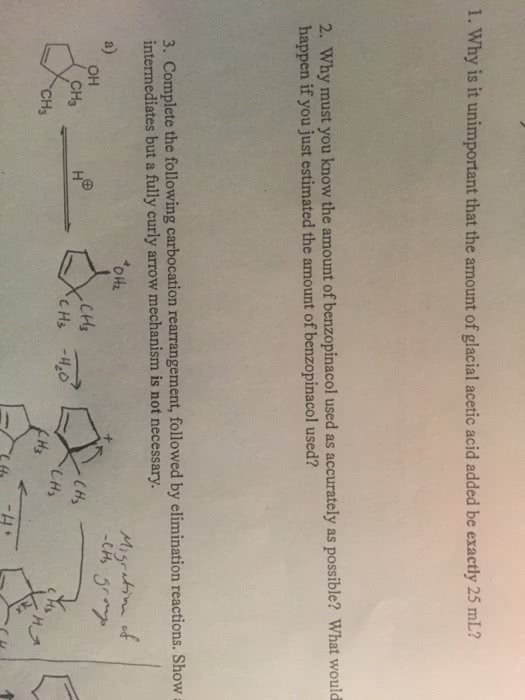
Questions 1 & 2 I need help with if possible?

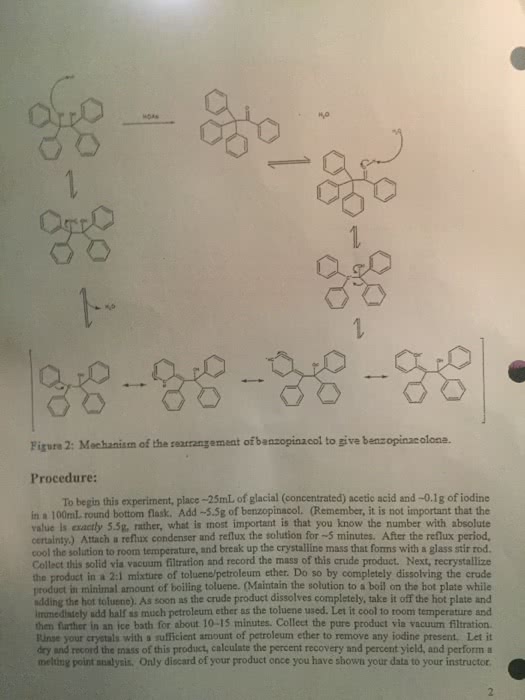
Synthesis of Benzopinacolone Purpose of the Experiment: esize benzopinacolone from benzopinacol in a The purpose of this week's experiment is to synth one-step rearrangement reaction. Background Information: Rearrangement reactions are a double edged sword in chemistry. Sometimes they can be helpful, and lead to the desired product as in today's reaction. In other times they can be harmful whereby carbon skeleton rearrangements lead to a plethora of products, not one of which may be desired. Today, we are going to use acid catalysis to rearrange benzopinacol to form benzopinacolone figure 1. Figure 1: Reaction of benzopinacol with acid to give the rearranged product, benzopinacotone. Figure 2 below describes the complete mechanism for the formation of benzopinacolone under acidic conditions. In an acidic environment, one of the becomes protonated to give a good leaving group, water. The water then leaves leaving a resonance stabilized carbocation. This carbocation is quite stable due to the fact that it is tertiary, as well as resonance stabilized by both benzene rings. In the next step of the mechanism one of the neighboring benzene rings shifts along with the concomitant formation of a carbon-oxygen pi bond. In the last step of the mechanism, the newly formed ketone is deprotonated by solvent or the conjugate base of the acid, and the product is