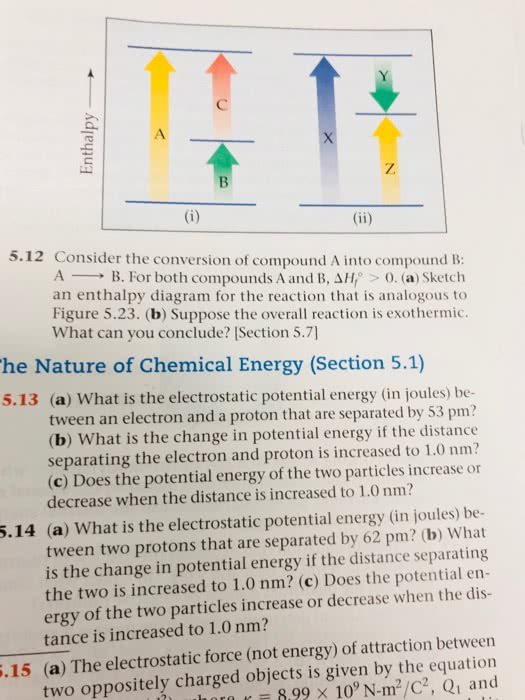
CHEM 121 Chapter : Chem 121 Chapter 5 Notes

79
CHEM 121 Full Course Notes
Verified Note
79 documents
Document Summary
Coulomb "s law : potential energy of attraction between two charges. Applied to quantify attraction between nucleus and electron, * Vcr / %9 r charge of nucleus - e winter) : potential energy. 91 : charge of particle one : proportional q, : charge of particle two r : distance between particles sign. As r decreases , magnitude of potential energy cvi increases. Forces between two charged particles (cid:8869) stronger at short distances , weaker at longer oppositely charged particles : attractive forces. Lowest / most negative) pe will occur at short distances - small r similarly charged particles : repulsive forces. Lowest pe occurs at long distances - larger. Potential energy /v7 only depends on r. Use spherical polar coordinates for wavefunctions (cid:8869) atoms have spherical shapes r , q , Variable r is distance from point of interest to the origin erector. D- is angle between f and z-axis.