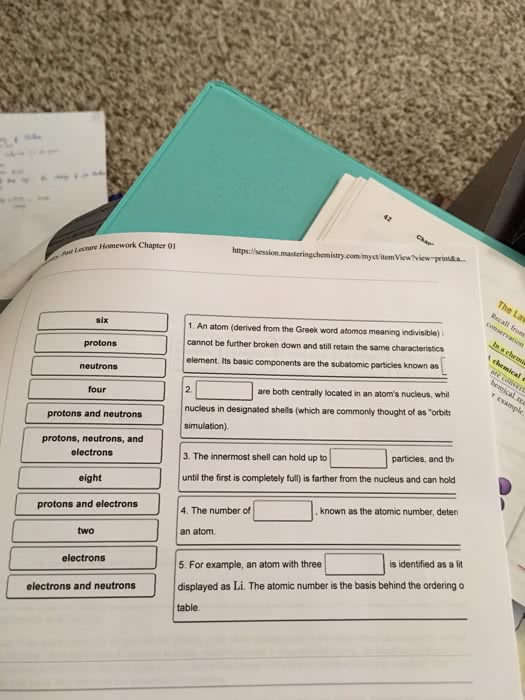
L07 Chem 111A Study Guide - Midterm Guide: Bohr Radius, Photon, Matter Wave
Document Summary
Chemistry 111, fall 2017, study guide, exam 1. Electric field (e: f=qe, force of electric field=charge of electron*magnitude of electric field, columbic force: forces exerted from space, electric field lines always point towards the negative charge and away from the positive charge. If a positive charge moves into an electric field it will experience a force and acceleration in the same direction of the field lines. If a negative charge moves into an electric field it will experience force and acceleration in the opposite direction of the field lines. Magnetic field (h or b: moving charged particles also experience a force in magnitude, magnitude of magnetic field is measurable and can be constant. Ke increases: f(r)= (cid:3044)(cid:3117)(cid:3044)(cid:3118) (cid:2872)(cid:3116)(cid:3045)(cid:3118) (measured in newton"s) If f(r) < 0 =attractive: the slope of force will be more drastic then the slope of potential energy. Determination of structure of atom (experiments & models)