CHEM 1127Q Lecture Notes - Lecture 2: Unified Atomic Mass Unit, Atomic Number, Periodic Table

Chapter 2 Atoms, Molecules, and Ions
Atomic mass unit (amu)
A (Angstrom)
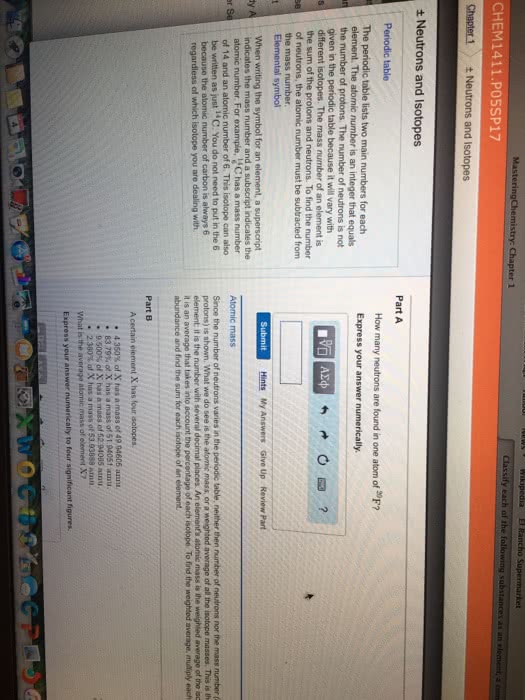
Atomic numbers is represented by Z, number of protons in an element
Mass Number(A)- numbers of protons and numbers of neutrons
A=Z # of Neutrons
A-Z= # of Neutrons
Numbers of protons= the number of electrons
Protons= 11 (same as atomic)
Neutrons=12 mass-atomic
electrons= 11(same as protons)
Number of protons can stay the same but the number of neutrons can change called isotopes
Isotopes- atoms of an element with the same number of protons but different numbers of neutrons or
atoms with the same atomic number but different mass #
12/6C 13/6C- symbols have to be the same
12/6C 13/6N- can not happen
Isobars- Atoms with the same mass # but different atomic number
Ca-41 K-41
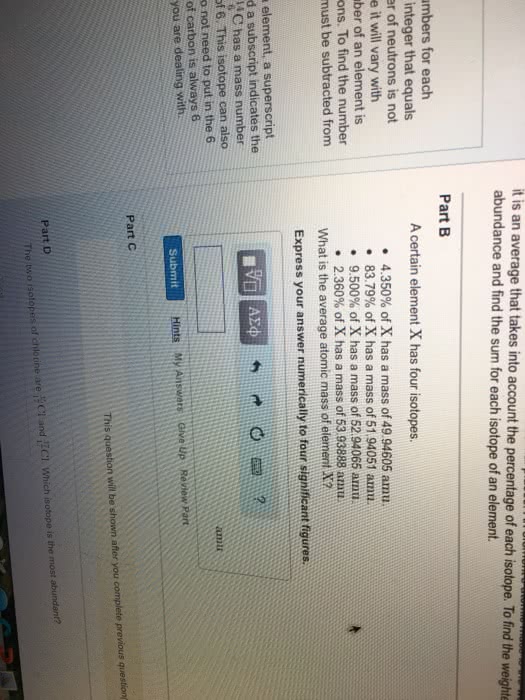
Atomic mass of element A= (atomic mass of A1/100+ (atomic mass of A2% A2/100+
Example: Find the atomic mass of CL
Atomic mass Abundance
C-35 34.97 75.33%
C-37 36.67 24.47%
Atomic mass of CL= (34.97) 75.53/100+(36.97)24.47/100= 35.46 amu
Periodic Table
Vertical lines= Groups
find more resources at oneclass.com
find more resources at oneclass.com

44
CHEM 1127Q Full Course Notes
Verified Note
44 documents