CHEM 1124Q Lecture Notes - Lecture 12: Nitride, Intravenous Sodium Bicarbonate, Dry Ice
67 views5 pages
26 Mar 2018
School
Department
Course
Professor
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related Documents
Related Questions
What is the pH of a solution made by dissolving 4.3 g of K2CO3 and 1.6 g of KHCO3 in 500. mL of water? (See the appendix.)
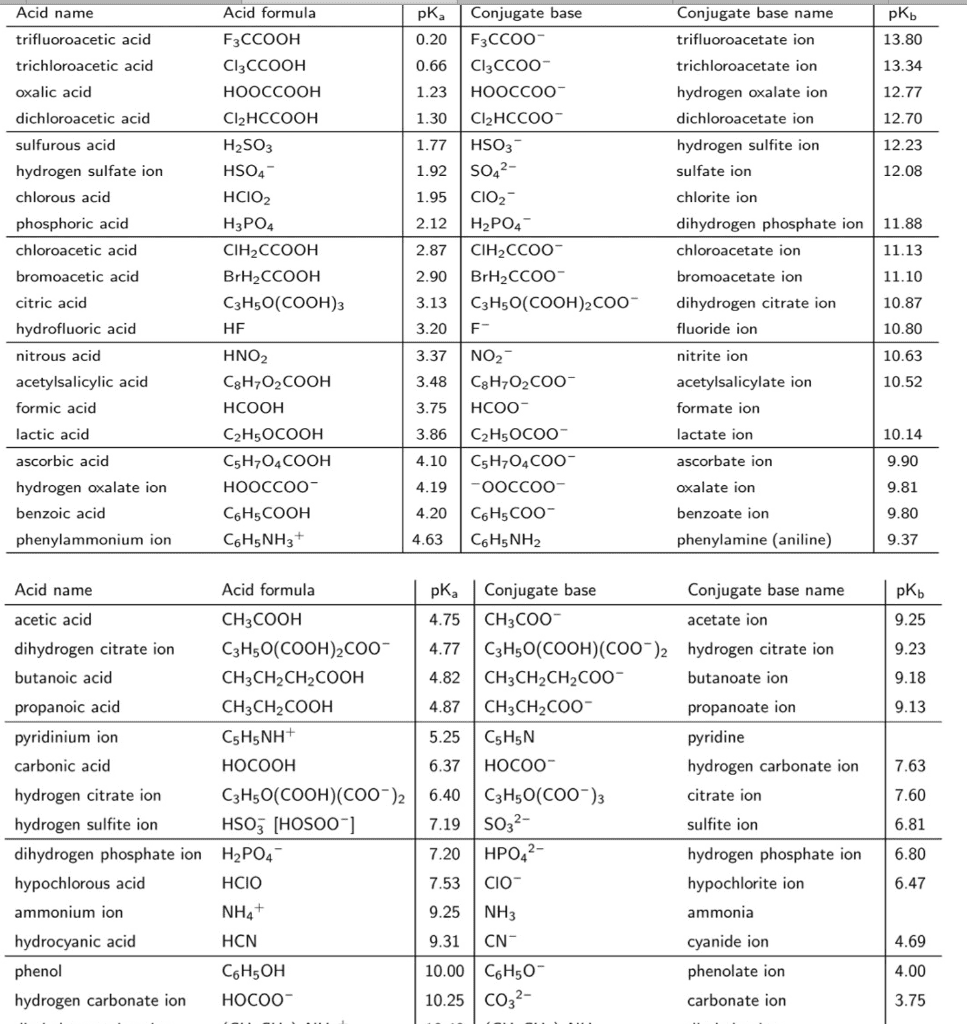
| Table 3 An Acid-base table | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 15.5:
| Name od Acid | Formula | Ka | Conjugate Base | Kb |
| Carbonic acid | H2CO3 | 4.2x10-7 | HCO3- | 2.4x10-8 |
| Hydrogen Carbonic Acid | HCO3- | 4.8x10-11 | CO32- | 2.1x10-4 |
| Sulfuric Acid | H2SO4 | very large | HSO4- | very small |
| Hydrogen Sulfate Ion | HSO4- | 1.3x10-2 | SO42- | 7.7x10-13 |
| Phosphoric Acid | H3PO4 | 7.5x10-3 | H2PO4- | 1.3x10-12 |
| Dihydrogen Phosphate ion | H2PO4- | 6.2x10-8 | HPO42- | 1.6x10-7 |
| Hydrogen Phosphate Ion | HPO42- | 4.8x10-13 | PO43- | 2.1x10-2 |
1. See table 15.5 for acid dissociation constants. What are the concentrations of H+ and CO32- respectively for a 0.073 M carbonic acid solution? Assume there is no loss of any carbonate species via generation of CO2
2,See table 15.5 for acid dissociation constants. What are the concentrations for H+ and SO42- respectively for a 0.15 M sulfuric acid solution?
3.See table 15.5 for acid dissociation constants. What are the concentrations of H+ and PO43- respectively for a 0.15 M phosphoric acid solution?