CHE 110L Lecture Notes - Lecture 3: Great Salt Lake
Document Summary
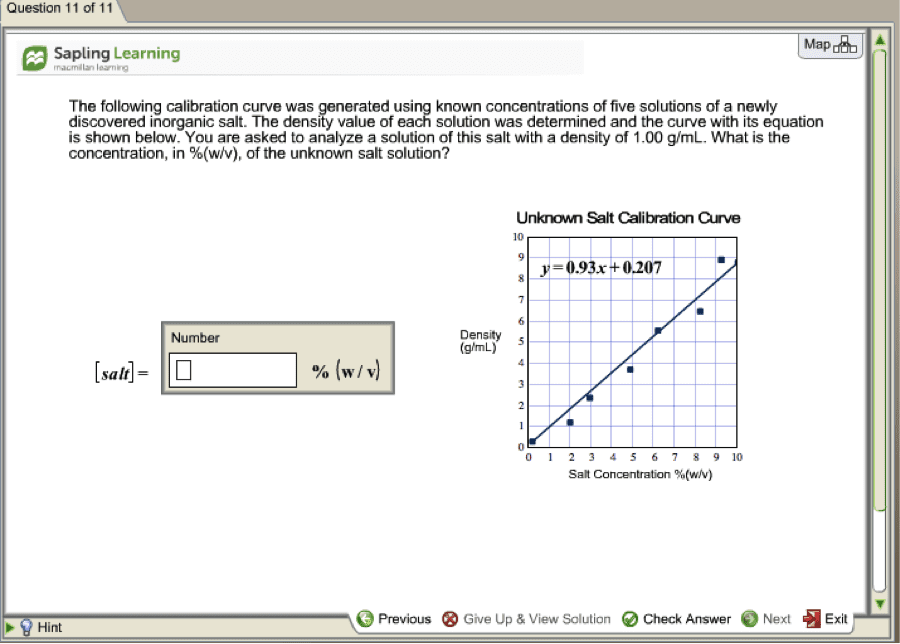
The procedure for this experiment can be found on pages 40-44 in general chemistry. Che110l laboratory manual fall 2016. the data for solution 4 was given in class due to the lack of time. Carbon copies of the data table have been attached to the back of this lab report. 48. 517g x 100 = 14. 26: % salt in, solution 3, solution 2: grams of salt grams of salt. 6. 921g grams of solution = grams of solution = volume (cid:4666)ml(cid:4667) = 3: in solution 2, in solution 1: 1. 046g+1. 048g+1. 046g: density of salt solution 1, average density: = 1. 021g/ml: slope: using the points (4,1. 024) and (14,1092) (1. 092g/ml-1. 024g/ml) = 0. 0068 g/ml: unknown 1: x, unknown 2: x= = 3. 56% (cid:3051)(cid:3118)-(cid:3051)(cid:3117) = m = (cid:3052)(cid:3118)-(cid:3052)(cid:3117: y intercept: y=mx+b. The purpose of this lab was to determine the concentration of salt in unknown salt solutions by plotting the linear relationship between density and concentration of standard salt solutions.