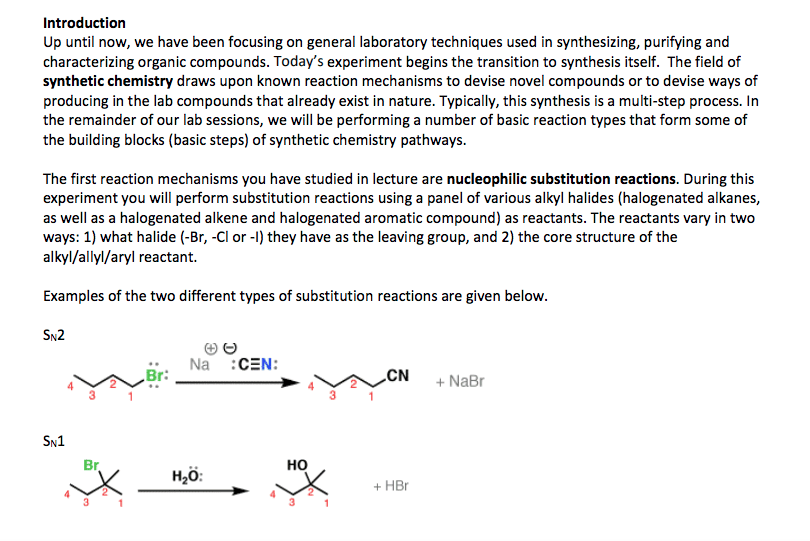
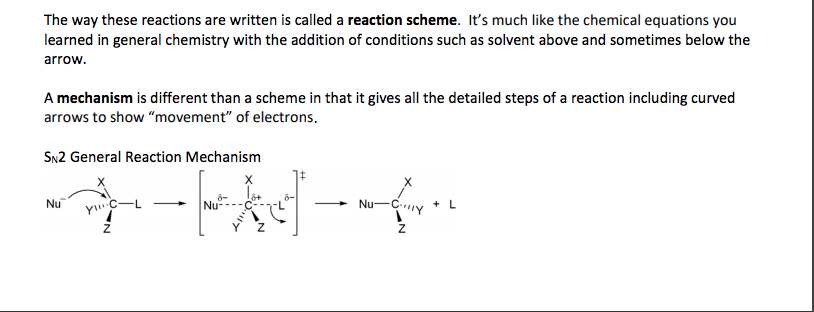
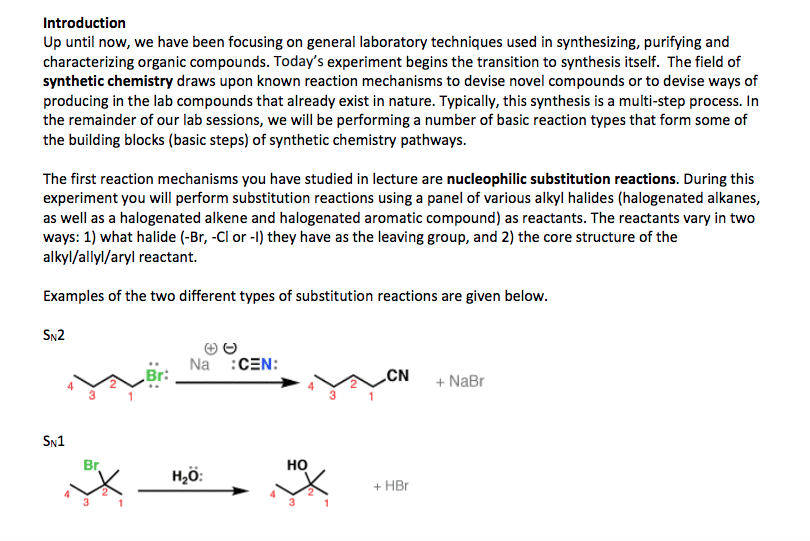
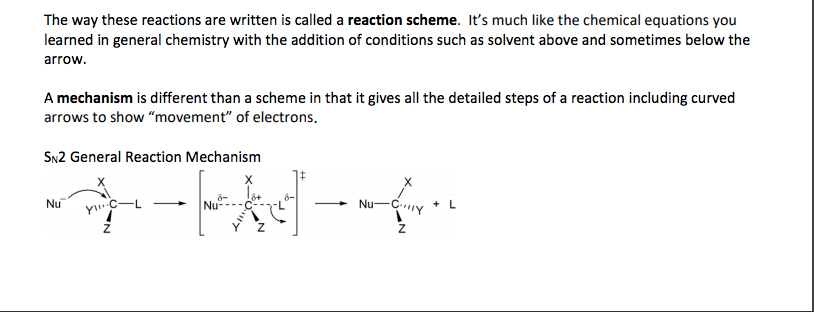
Chemistry CHEM S-20ab Lecture Notes - Lecture 8: Racemic Mixture, Diastereomer, Stereoisomerism


Chemistry E-2a: Lecture 5 October 23, 2008
12
Stereoisomers as Reactants
• We are often interested in the differences in reactivity of various chemical compounds.
Usually, we express the differences in reactivity in terms of the rates of the reactions.
How is the rate of a reaction related to the energies of the reactant, the transition state,
and the product?
• We can make several generalizations about rates of reactions involving various
stereoisomers. Can you explain why each of these generalizations is true?
Compounds that are diastereomers may react at different rates with any reagents.
Compounds that are enantiomers must react at the same rate with achiral reagents.
Compounds that are enantiomers may react at different rates with chiral reagents.
Reading: Section 7.7
Chemistry S-20ab
Week 2
91

Chemistry E-2a: Lecture 5 October 23, 2008
13
Stereoisomers as Products
• Determine the product of the following reaction:
H2 , Pt
• Is the product chiral or achiral? Are the reactants and reagents chiral or achiral?
• We can make several generalizations about reactions that form stereoisomeric products.
Can you explain why each of these generalizations is true?
Achiral starting materials that give enantiomeric products will yield a racemic mixture.
Achiral starting materials that react in a chiral environment (e.g. a chiral catalyst or chiral
solvent) may yield different amounts of each enantiomer.
Achiral starting materials that give diastereomeric products may yield different amounts
of each diastereomer.
Reading: Section 7.8
Chemistry S-20ab
Week 2
92
Document Summary
We are often interested in the differences in reactivity of various chemical compounds. Usually, we express the differences in reactivity in terms of the rates of the reactions. We can make several generalizations about rates of reactions involving various stereoisomers. Compounds that are diastereomers may react at different rates with any reagents. Compounds that are enantiomers must react at the same rate with achiral reagents. Compounds that are enantiomers may react at different rates with chiral reagents. We can make several generalizations about reactions that form stereoisomeric products. Achiral starting materials that give enantiomeric products will yield a racemic mixture. Achiral starting materials that react in a chiral environment (e. g. a chiral catalyst or chiral solvent) may yield different amounts of each enantiomer. Achiral starting materials that give diastereomeric products may yield different amounts of each diastereomer. The most common ring in natural compounds is a 6-membered ring. Take out your model kits and make a model of cyclohexane: