CHE 331 Chapter Notes - Chapter 5: Stereocenter, Enantiomeric Excess, Specific Rotation
Document Summary
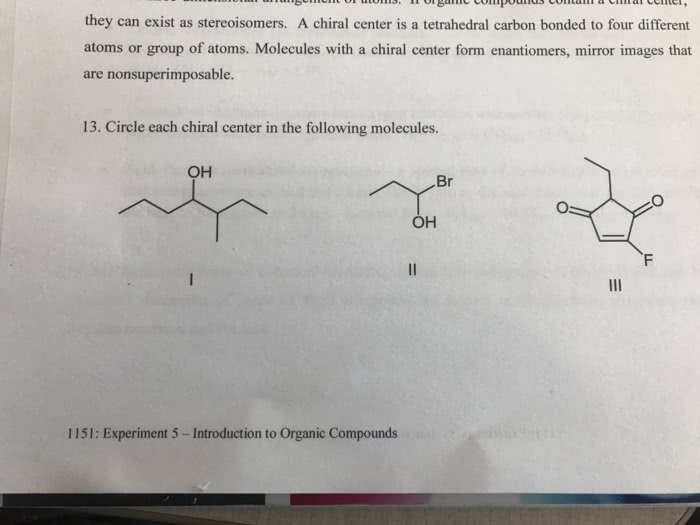
They must be tetrahedral: a molecule will not be chiral if it possesses a plane of symmetry. A plane of symmetry (also called a mirror plane) is defined as an imaginary plane that bisects a molecule in such a way that the two halves of the molecule are mirror images of each other. The plane may pass through atoms, between atoms, or both: according to the r,s-system, one enantiomer of 2-butanol should be designated (r)-2-butanol and the other enantiomer should be designated (s)-2-butanol. [(r) and (s) are from the latin words rectus and sinister, meaning right and left, respectively. ] These molecules are said to have opposite configurations at c2: each of the four groups attached to the chirality center is assigned a priority. Priority is determined by higher atomic number (of every connecting atom). If 2 atoms are the same, look at their next set of attached atoms.