CHEM 102 Lecture Notes - Lecture 4: Elimination Reaction, Leaving Group, Molecularity
85 views2 pages
Document Summary
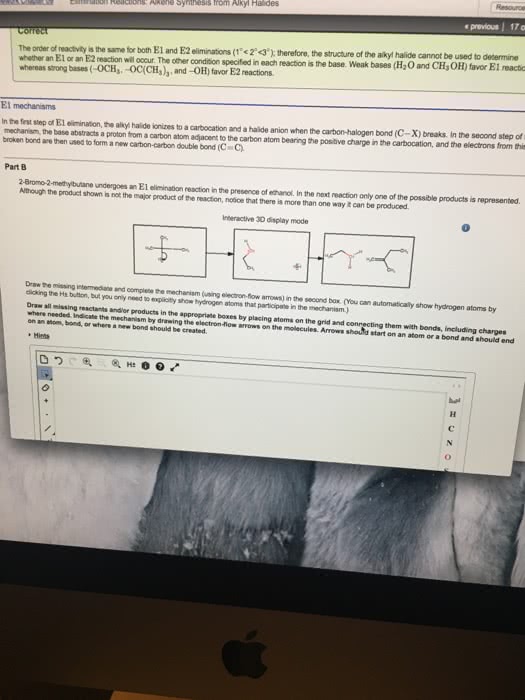
There is two mechanisms for elimination: e1 describes an elimination reaction (e) in which the rate-determining step is unimolecular (1) and does not involve the base. The leaving group leaves in this step, and the proton is removed in a separate second step: e2 describes an elimination (e) that has a bimolecular (2) rate-determining step that must involve the base. Loss of the leaving group is simultaneous with removal of the proton by the base. There are a number of factors that affect whether an elimination goes by an e1 or e2 mechanism. One is immediately obvious from the rate equations: only the e2 is affected by the concentration of base, so at high base concentration e2 is favoured. If you see a strong base being used for an elimination, it is certainly an e2 reaction. We have already mentioned the bulky t-butoxide ideal for promoting e2 as it"s both bulky and a strong base (pkah = 18).
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232