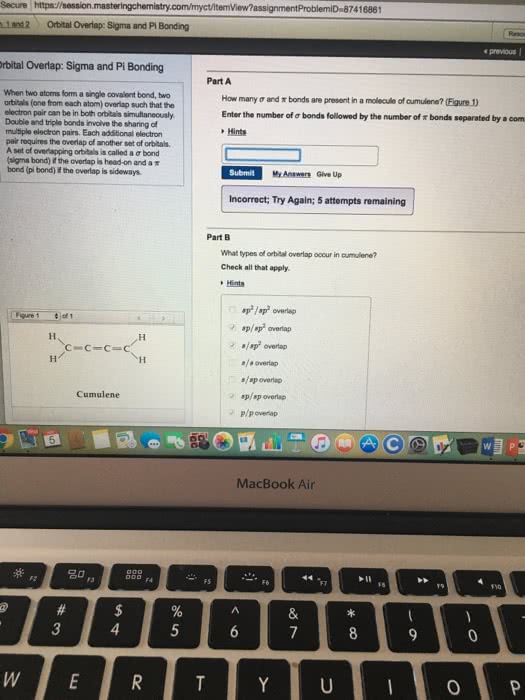
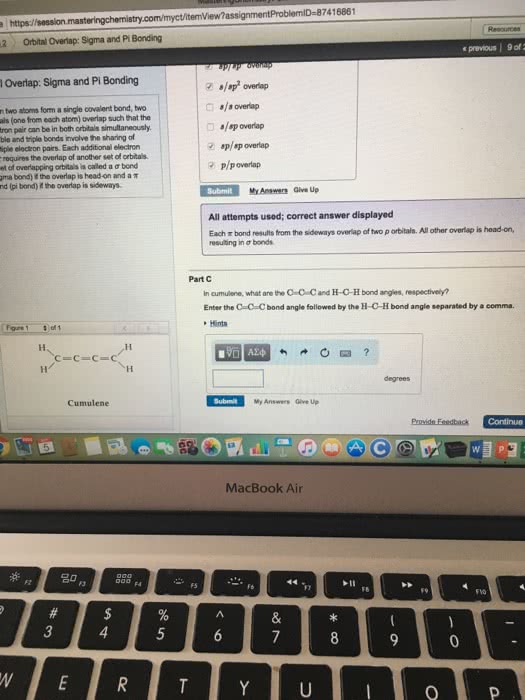
7416861 Secure https://session Ln2 Obital Overlap: Sigma and Pi Bonding e previous rbital Overlap: Sigma and Pi Bonding Part A When two atoms form a single covalent bond, two orbitals (one from each atom) overlap such that the electron pair can be in both orbitais simultaneously Double and triple bonds invoive the sharing of multiple electron pairs. Each additional electron pair requires the overlap of another set of orbitals Aset of ovenapping ort tals is called a Ï bond (signa bond) if the overlapishead-on and a Ï bond (pi bond) i the overlap is sideways How many Ï and Ï bonds are present in a molecto ofcumulene? Pam) Enter the number of Ï bonds followed by the number of Ï bonds separated by a com Hints Submit My Answers Give Up Incorrect; Try Again; 5 attempts remaining Part B What types of orbital overlap occur in oumulene? Check all that apply of 1 ./sp2 overlap /s overlap s/sp overlap sp/sp overlap Cumulene MacBook Air 20 F6 0