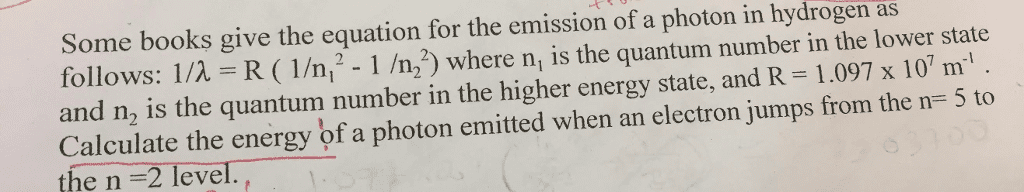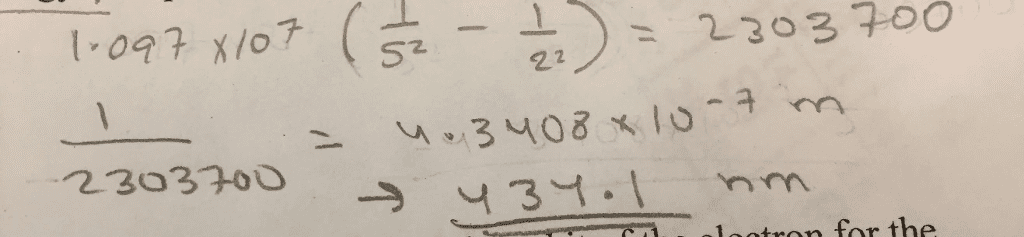CHM135H1 Lecture Notes - Lecture 5: Ableton, Bohr Model, Kilogram

90
CHM135H1 Full Course Notes
Verified Note
90 documents
Document Summary
Chm135h1 f - lecture 5 - chapter 5 (3) At n = (i. e. ionization energy), the electron becomes unbound. Cons: only applies to one-electron atoms, e. g. h, he+, li2: can"t explain interactions between electrons, applies to a very limited type of atoms / ions. The balmer-rydberg equation is used to calculate the energy of electron transitions in the hydrogen atom. * will have to change units of r if is in m. Matter also has both particle and wave properties (like electromagnetic radiation) We know that e = hc and that the energy of a certain mass is e = mc2. We can combine the two equations, substituting c with v for velocity (since we don"t travel at the speed of light) hv. Example 4: what is the wavelength of a 0. 11 x 10-31 kg baseball moving at 1% of the speed of light? (h = 6. 626 x 10-34 j s; 1 j = 1 kg m2 s-2) h mv.