CHMA10H3 Lecture Notes - Lecture 3: Molar Mass, Limiting Reagent, Reagent
208 views3 pages
Verified Note
10 Sep 2018
School
Department
Course
CHMA10H3 verified notes
3/39View all
Get access
Grade+
$40 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
10 Verified Answers
Class+
$30 USD/m
Billed monthly

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
7 Verified Answers
Related Documents
Related Questions
Why must chemical equations be balanced?
Choose all that apply.
Choose all that apply.
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the given reaction is possible or not. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict whether the reaction requires a catalyst or not. | |
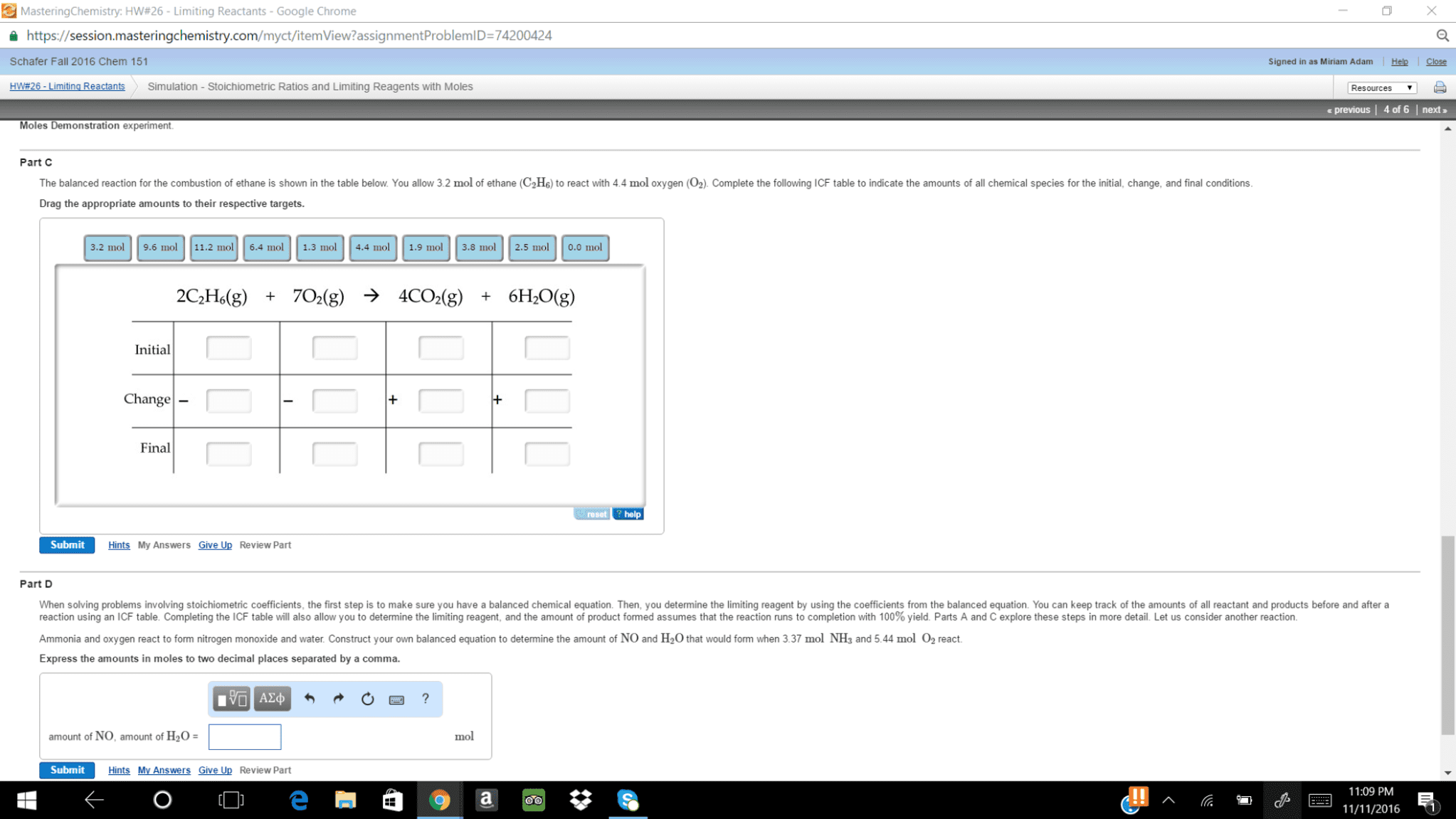
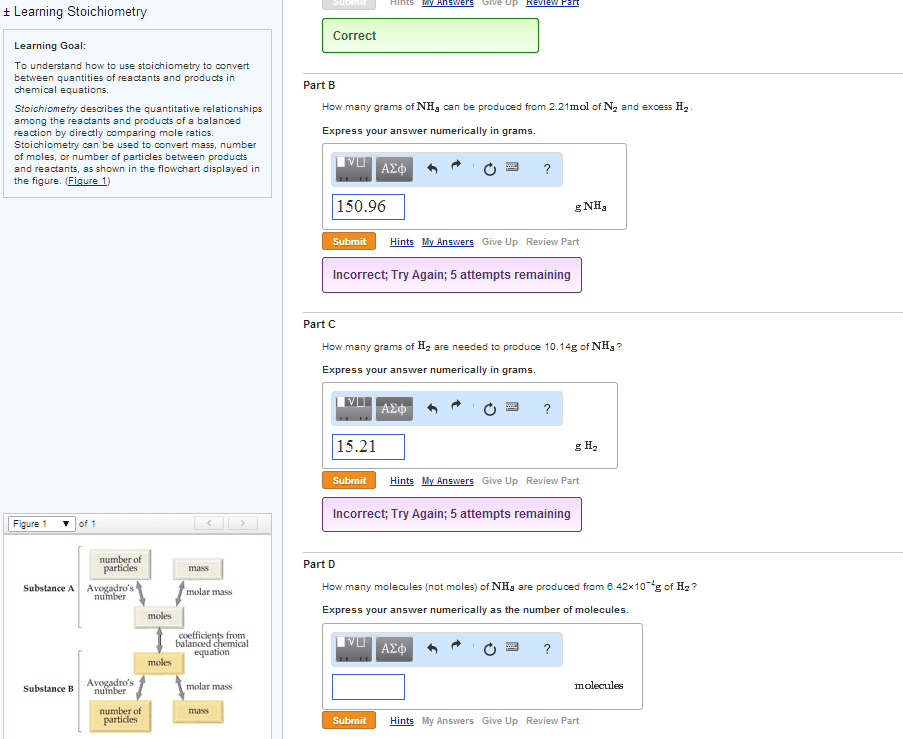
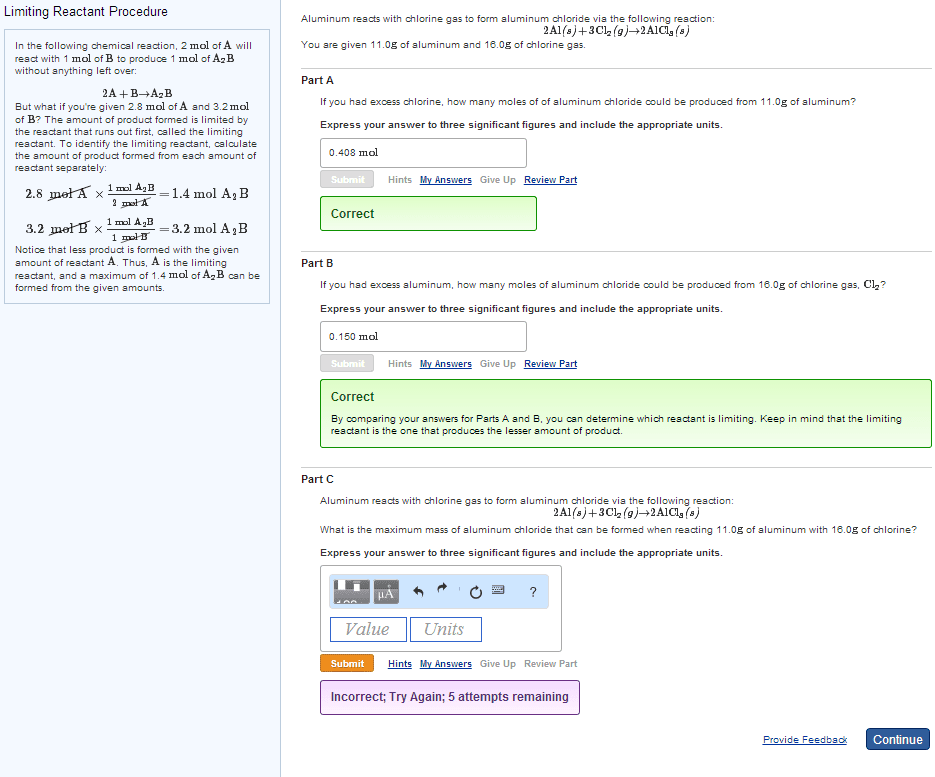
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict the amount of product that can form from a given amount of reactant. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much of one reactant is required to react with a given amount of another. | |
| In a balanced chemical equation, the coefficients in front of each reactant and product allow us to predict how much reactants are required to form a given amount of products. |