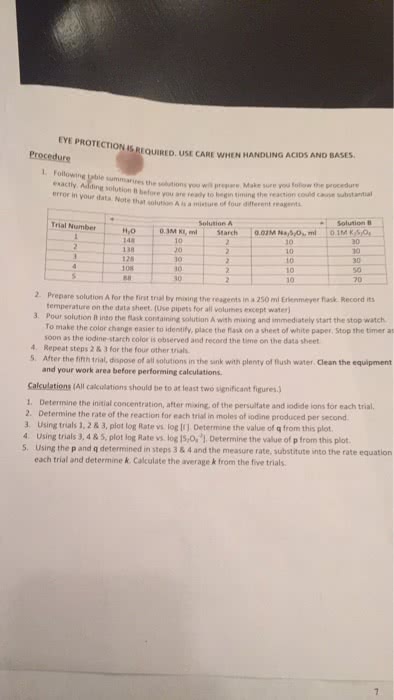
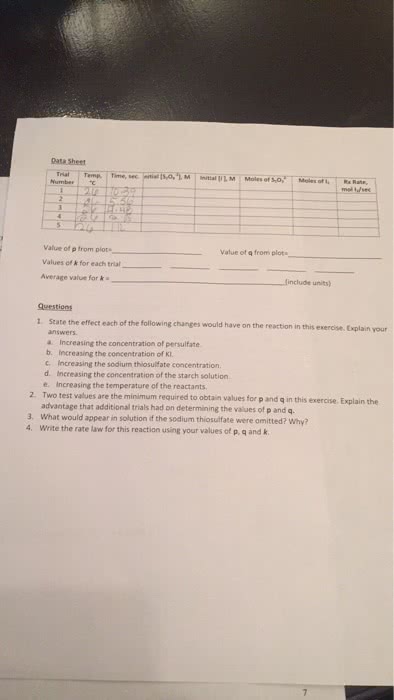
CHM 2123 Lecture Notes - Lecture 2: Bromothymol Blue, Leaving Group, Stereocenter
Document Summary
Experiment 2 the kinetic study of nucleophilic substitution. Possible way to optimize yield observe kinetics of reaction --> postulate a mechanism. Observed by rate of disappearance of reactants or rate of formation of products. A + b --> c + d is. X and y represent the order of the reaction with respect to each reactant (determined experimentally) Sum of these powers determine overall order of the reaction. K is the rate constant of the reaction, depending on experimental conditions. Usually a rxn is zero order for one reactant but a higher order for another. Formation of products can only dependant on the concentration of one sm. Graph of [reactants] vs time, is linear and with a slope of k (rate constant) Graph of ln[reactants] vs time has slope of - k. Graph of 1/[reactants] vs time has slope of k. [a] = [a]0e -kt ln[a]t = ln[a]0 kt.