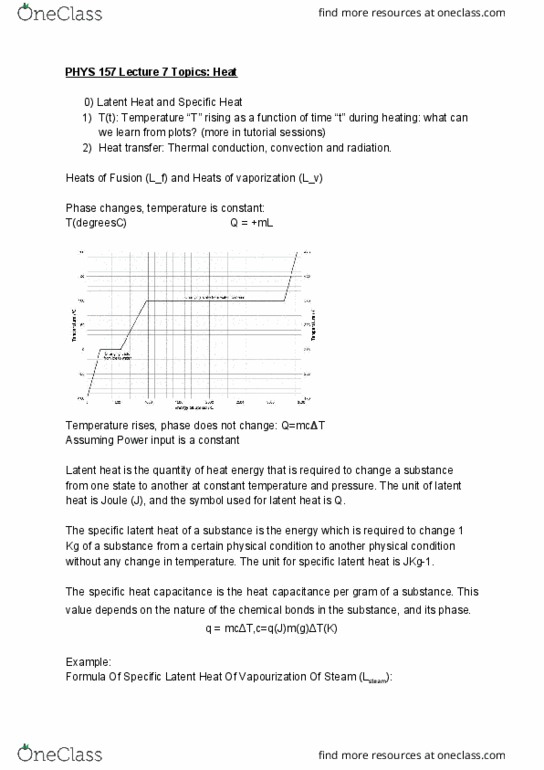
PHYS 157 Lecture 6: PHYS 157 Lecture 6
PHYS 157 verified notes
6/26View all
Document Summary
The pressure exerted on a wall of a container of gas is due to the constant bombardment of molecules. If the volume is reduced by half, the molecules are closer together and twice as many will be striking a given area of the per second. Hence he would expect the pressure to be twice as great. Magnitude of the force (according to newton"s second law is equal to the molecule"s rate of change of momentum. The variable is the x component of the molecule that changes. Thus the change in momentum change is the final minus the initial: The average force - averaged over many collisions - will be equal to the force exerted during one collision divided by the time between collisions (newton"s second law)