CHEM 1050 Lecture Notes - Lecture 11: Elementary Reaction, John Wiley & Sons, Collision Theory
Document Summary
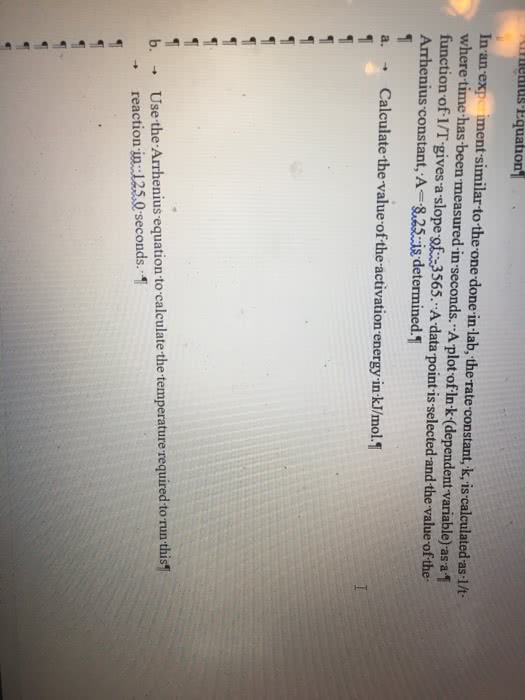
Experimentally ( increases / decreases ) as the temperature is raised. for most found it is that reactions, the reaction rate. By trying various combinations of plots, it was soon realised that a plot of ln k vs. 1/t (or log k versus 1/t) gave a linear plot with a negative slope. Arrhenius supplied the answer in 1899: k = a exp{ ea/rt} where k = rate constant, a = constant and ea = activation energy. The arrhenius equation allows us to do is to calculate the activation energy for a reaction if we know the rate constants for the reaction at two temperatures. Thus: (1) at t2: ln k2 = ln a (ea/r)(1/t2) (2) at t1: ln k1 = ln a (ea/r)(1/t1) Subtract equation (1) from (2): ln (k2/k1) = (ea/r)(1/t2 1/t1) By rearranging the equation: ln (k2/k1) = (ea/r)(t1 t2)/(t1 t2) Or ln (k2/k1) = (ea/r)( t)/(t1 t2) know k at one temp, can find.