CHEM 1AA3 Lecture Notes - Equilibrium Constant, Potassium Thiocyanate, Thiocyanate
Document Summary
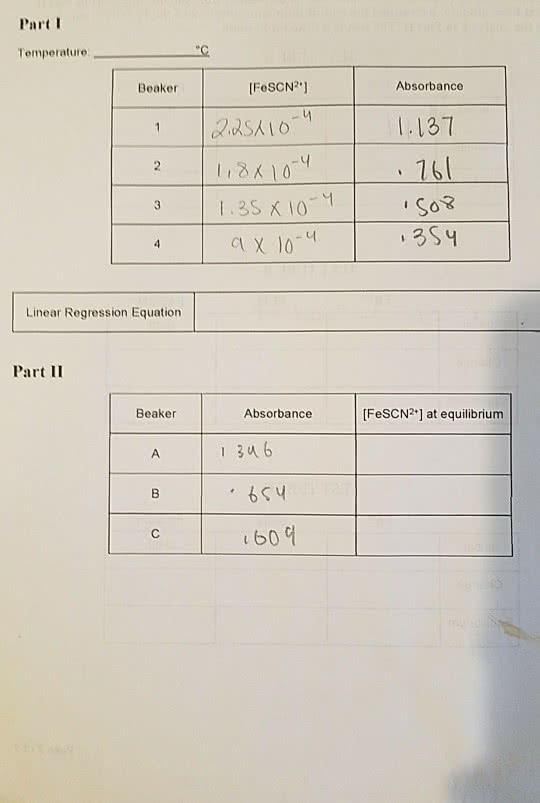
To find the equilibrium constant of the (kc) for the reaction: fe3+(aq) + scn-(aq) (aq) through several different concentrations of reactants, using spectrophotometric analysis. Please refer to the chem 1a03/1e03/1aa3 lab manual 2013, pages 43-47, for a detailed procedure. Table 1: values of absorbance and concentration for different volumes of potassium thiocyanate and 0. 200 mol/l ferric nitrate. Fescn2+(aq) and kscn (aq) were both clear, colourless liquids initially. Solution turned to an orange and brown colour when the ferric nitrate was added to the potassium thiocyanate. Fe3+(aq) + scn-(aq) n(scn-) = cv n(scn-) = (. 002 mol/l) (. 001(l)) n(scn-) = . 000002 mol. Due to 1:1 mol ratio, n(fescn2+) = . 000002mol. The above graph shows the proportional relationship between the concentration and the absrbance of fescn2+. Using a line of best fit, a proportional constant of 5223 is determined to be the slope. A = absorbance = 0. 243a y = proportionality constant = 5223 a/mol/l.