1) A solution is 1.00x10-4 M in SCN- an0.36 M in Fe3+ are mixed and react according to equationâ¨â¨
Fe3+(aq)+SCN-(aq)<--> (Fe(SCN))2+(aq)
Under these conditions, the equilibrium is driven all of the way tothe right. The absorbance of the solution at 447 nm is 0.500, andthe path length of the test tube is 1.00 cm. What is the value ofthe molar absorptivity, ε, for the complex FeSCN2+?
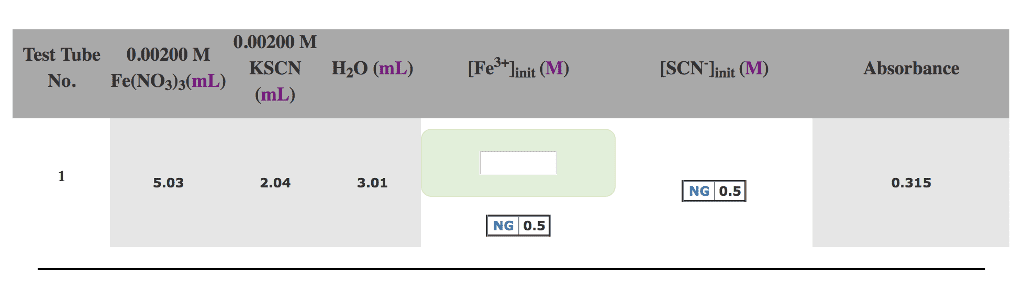
2) In a test tube, you combine the following:
2.00 mL of 0.00200 M Fe(NO3)3solution
2.00 mL of 0.00100 M KSCN solution
1.00 mL of 2.0 M HNO3 solution
3.00 mL of distilled water
a) What are the initial concentration of the reactants (i.e.,assuming no reaction occurs)?
(Fe3+)init = M
(SCN-)init= M
b) The absorbance of the resulting solution is 0.101 at 447 nm.What is the equilibrium concentration of the FeSCN2+product, based on the molar absorptivity determined above?
(FeSCN2+)equil= M
c) Set up an ICE table for the reaction, and determine the value ofKc from the data
Kc=
1) A solution is 1.00x10-4 M in SCN- an0.36 M in Fe3+ are mixed and react according to equationâ¨â¨
Fe3+(aq)+SCN-(aq)<--> (Fe(SCN))2+(aq)
Under these conditions, the equilibrium is driven all of the way tothe right. The absorbance of the solution at 447 nm is 0.500, andthe path length of the test tube is 1.00 cm. What is the value ofthe molar absorptivity, ε, for the complex FeSCN2+?
2) In a test tube, you combine the following:
2.00 mL of 0.00200 M Fe(NO3)3solution
2.00 mL of 0.00100 M KSCN solution
1.00 mL of 2.0 M HNO3 solution
3.00 mL of distilled water
a) What are the initial concentration of the reactants (i.e.,assuming no reaction occurs)?
(Fe3+)init = M
(SCN-)init= M
b) The absorbance of the resulting solution is 0.101 at 447 nm.What is the equilibrium concentration of the FeSCN2+product, based on the molar absorptivity determined above?
(FeSCN2+)equil= M
c) Set up an ICE table for the reaction, and determine the value ofKc from the data
Kc=