CHEM10006 Lecture Notes - Lecture 5: Wcen-Fm, Rice Chart, Gibbs Free Energy
36 views1 pages
10 Aug 2018
School
Department
Course
Professor
Document Summary
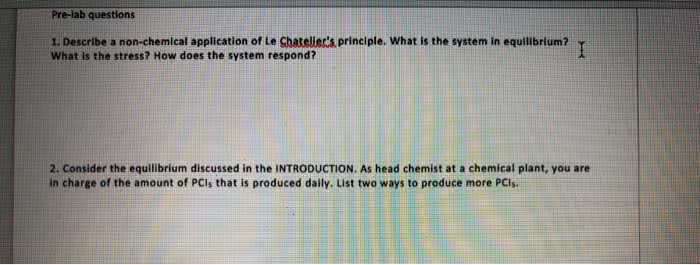
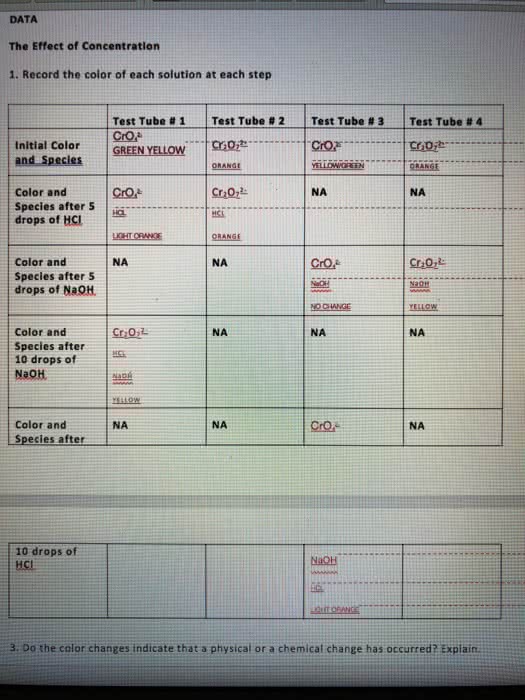
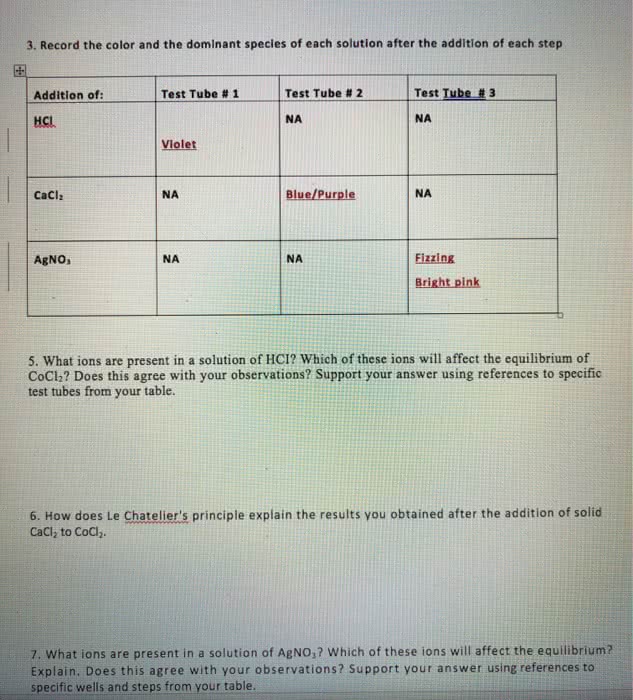
Gibbs energy is also the thermodynamic potential that is minimized when a system reaches chemical equilibrium at constant pressure and temperature. Used to calculate k at a specified temperature if another k value at a different temperature is known. (cid:2779) (cid:2778)= ((cid:2778)(cid:2778) (cid:2778)(cid:2779)) Catalysts affect the rates of chemical reactions without being used up. Addition of a catalyst may help bring a system to chemical equilibrium more rapidly. Addition of a catalyst does not affect the position of equilibrium. G, h, s and thus k not affe(cid:272)ted. The effects of temperature and catalysts on the equilibrium constant. Measure the concentrations of reactants and products after equilibrium reached. (ice table) Substitute these equilibrium values into the equilibrium constant expression to calculate kc. Not much reactants reacted (since kc is small) so assume x<
Get access
Grade+20% off
$8 USD/m$10 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
40 Verified Answers
Class+
$8 USD/m
Billed $96 USD annually

Homework Help
Study Guides
Textbook Solutions
Class Notes
Textbook Notes
Booster Class
30 Verified Answers
Related textbook solutions
Chemistry: Structure and Properties
2 Edition,
Tro
ISBN: 9780134293936
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232