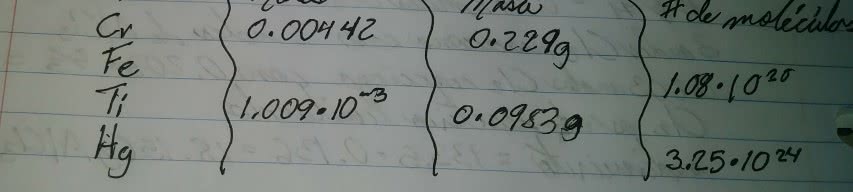CHEM101 Chapter Notes - Chapter 9: Cryoscopic Constant, Hydrophile, Colligative Properties
Document Summary
Solution a homogeneous mixture of two or more substances, in which one is called the solvent and the other is the solute. Solvent main medium, what is there in the largest quantity. Mass percent = mass solution/ total mass solution x 100% Molarity = moles solute/ total volume of solution. Useful for applications where the temperature (thus volume) of a solution changes. Defined as the number of moles of solute divided by the mass of the solvent in kg. Units are in mol/kg whereas molarity is mol/l. Miscible: when two liquids mix in all proportions (e. g. acetone and water) Immiscible: when two liquids do not mix at all (e. g. oil and water) Insoluble: when a solid does not dissolve in a solvent (nacl in gasoline) Saturated: a solution with the maximum possible solute concentration. 1. network solids (diamond, graphite) cannot dissolve without breaking covalent bonds. 3. metals do not dissolve in water (some will react, but not dissolve)