CHEM 1128Q Study Guide - Final Guide: Joule, Lithium Carbonate, Chemical Equation

19
CHEM 1128Q Full Course Notes
Verified Note
19 documents
Document Summary
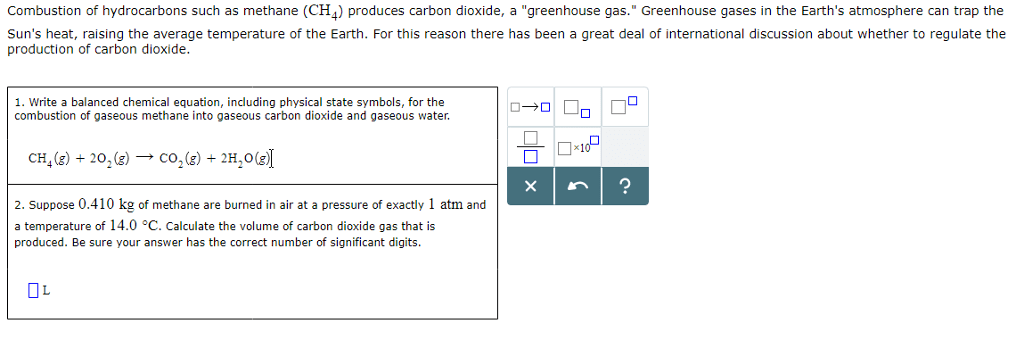
16. 3: the second and third laws of thermodynamics: write out the balanced chemical equation for the combustion of benzene, Ch4(g), to give carbon dioxide and water vapor. Explain why it is difficult to predict whether the change in entropy will be positive or negative. C6h6 (l), to give carbon dioxide and water vapor. Change in s is positive, because the moles of gas are increasing: determine the entropy change for the combustion of liquid ethanol, c2h5oh, under standard state conditions to give gaseous carbon dioxide and liquid water. No, it is not spontaneous at room temperature because the change in enthalpy value is positive, when it needs to be negative for a spontaneous reaction to occur at all temperatures. To find the temperature it is spontaneous at, you calculate the standard change in enthalpy divided by the standard change in entropy which gives you the minimum temperature the reaction is spontaneous at.