CHEM 1040 Study Guide - Midterm Guide: Bond-Dissociation Energy, Jmol, Joule
Document Summary
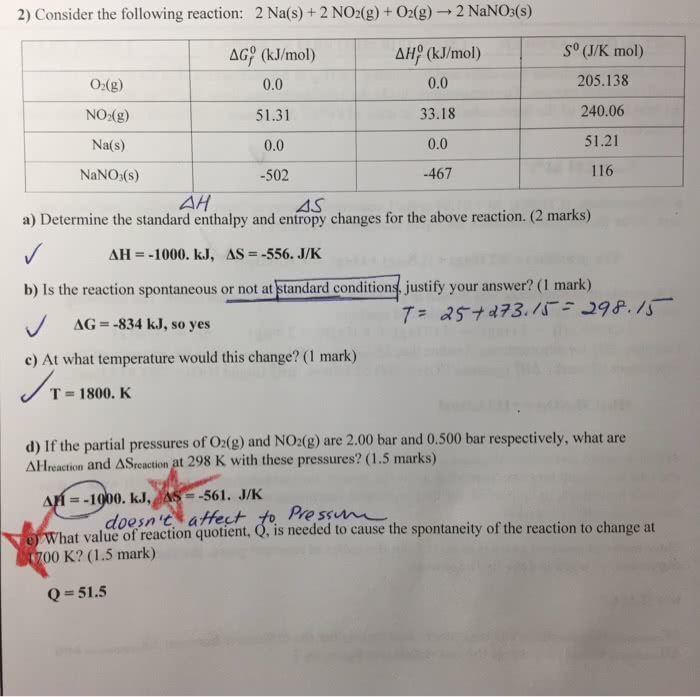
Write down the reaction equation, including phases, for the formation (under standard conditions) of 5-bromouracil (c4h3brn2o2), as a solid which is a molecule which can cause mutations in. 4 c(s) + 1. 5 h2(g) + 0. 5 br2(l) + n2(g) + o2(g) c4h3brn2o2(s) The bond enthalpy for the n n bond is 942 kj mol-1. Write down the chemical reaction equation for the molar enthalpy of formation for gaseous n atoms and give its value? (2 marks)! What is the sign of the entropy change in each of these reactions. F2(g) 2 f(g) positive negative no change! C3h8(g) + 5 o2(g) 3 co2(g) + 4 h2o(l) positive negative no change! Not marking sig gs unless really bad (more than 2 places)! Nitrogen dioxide decomposes according to the following equation: The data below each species is its standard molar free energy of formation at 298 k. What is the standard molar free energy of reaction? (2 marks) (i)