11
answers
0
watching
54
views
15 May 2023
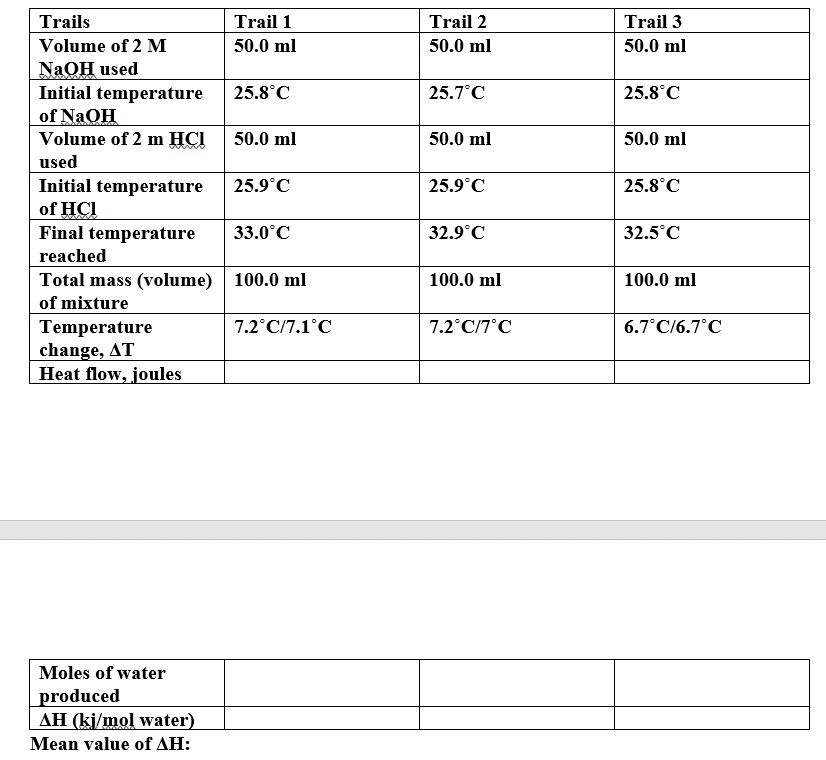
Given: 1 M HCl (30mL), 1 M NaOH (30mL), total volume of 60 mL, initial temperature of 19.1 °C, final temperature of 25.8 °C.
Solve for:
1. Initial mol H+ (mol)
2. Initial mol OH- (mol)
3. mol H2O produced (mol)
4. ÎT, °C
5. qneut (J)
6. ÎHneut, (kj/mol)
Given: 1 M HCl (30mL), 1 M NaOH (30mL), total volume of 60 mL, initial temperature of 19.1 °C, final temperature of 25.8 °C.
Solve for:
1. Initial mol H+ (mol)
2. Initial mol OH- (mol)
3. mol H2O produced (mol)
4. ÎT, °C
5. qneut (J)
6. ÎHneut, (kj/mol)
karimbalticLv7
4 Nov 2023
Unlock all answers
Get 1 free homework help answer.
Already have an account? Log in
wahabmunir796Lv10
11 Jul 2023
Get unlimited access
Already have an account? Log in
20 May 2023
Get unlimited access
Already have an account? Log in
esskeetitLv10
17 May 2023
Get unlimited access
Already have an account? Log in
exeq2001Lv3
16 May 2023
Get unlimited access
Already have an account? Log in
OC4883347Lv10
16 May 2023
Get unlimited access
Already have an account? Log in
nguyenngocyLv10
16 May 2023
Get unlimited access
Already have an account? Log in
16 May 2023
Get unlimited access
Already have an account? Log in
gnana0537Lv10
16 May 2023
Get unlimited access
Already have an account? Log in
Tod ThielLv2
16 May 2023
Get unlimited access
Already have an account? Log in
dqvinh05Lv10
16 May 2023
Get unlimited access
Already have an account? Log in