1
answer
0
watching
262
views
16 Nov 2019
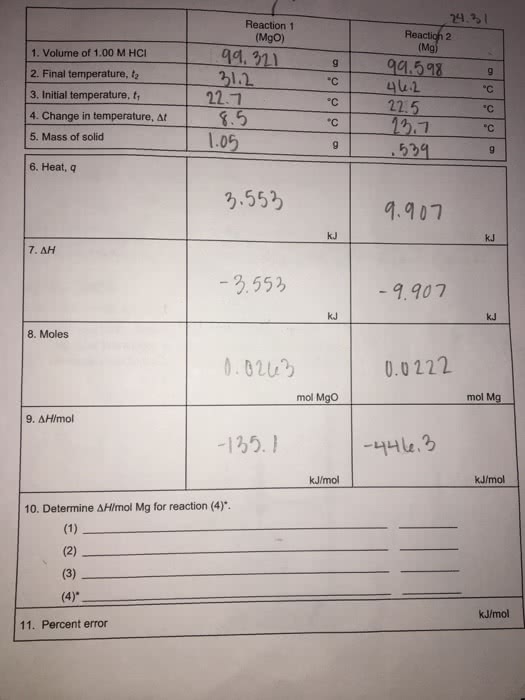
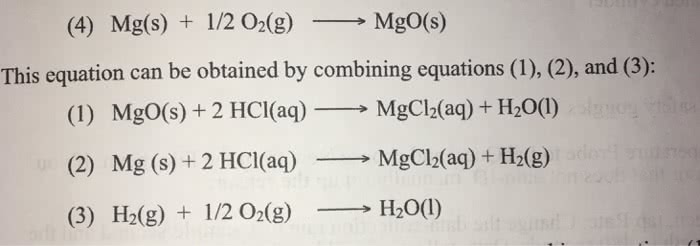

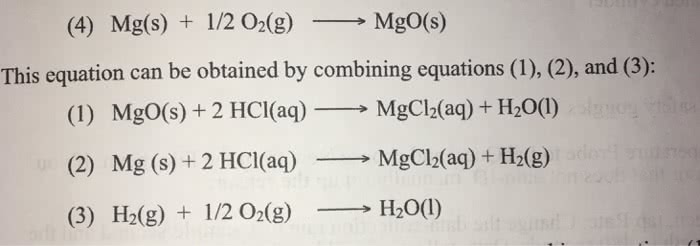
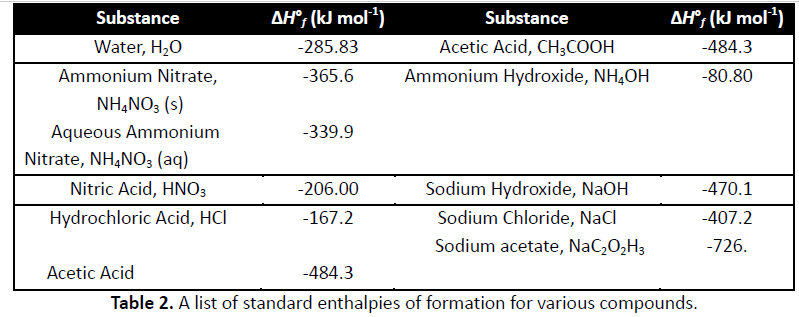
Reaction 1 (MgO) Reactioh 2 (Mg valume of 1,00 M HC1 94, 911 11 5.5 1.05 2. Final temperature, t2 3. Initial temperature, t 41.2 22.5 4. Change in temperature, At 5. Mass of solid °C 95299 6. Heat, q .553 9.907 kJ 23.55 - 9.907 KJ 8. Moles 0.0222 mol MgO mol Mg 135. kJ/mol 10. Determine ÎHmol Mg for reaction (4)". kJ/mol 11. Percent error
1
answer
0
watching
262
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Collen VonLv2
9 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232