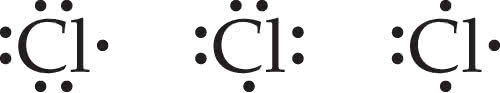2
answers
1
watching
718
views
28 Nov 2020
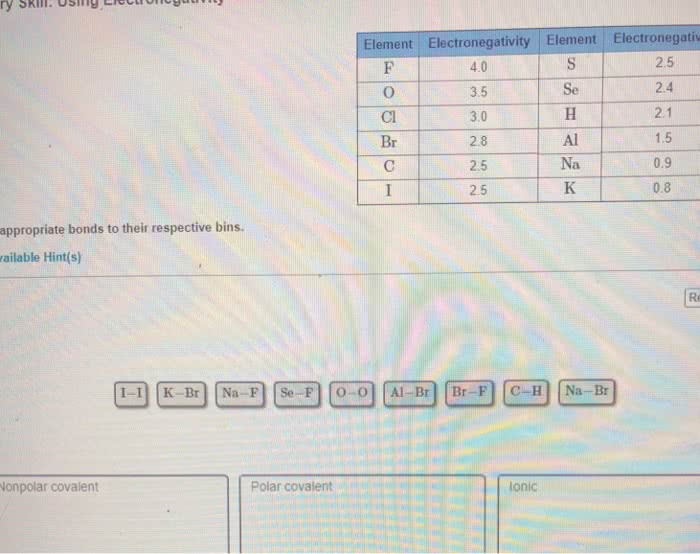
Classify the bonds formed between each pair of elements based on electronegativity values in the following table:
Element
Sc
Be
H
P
S
I
Br
Cl
O
F
Electronegativity
1.3
1.5
2.1
2.1
2.5
2.5
2.8
3.0
3.5
4.0
Drag the appropriate items to their respective bins.
Ionic
polar covalent
non-polar covalent
S-I
H-Br
H-P
S-O
Be-F
Be-Cl
Sc-O
Classify the bonds formed between each pair of elements based on electronegativity values in the following table:
| Element | Sc | Be | H | P | S | I | Br | Cl | O | F |
| Electronegativity | 1.3 | 1.5 | 2.1 | 2.1 | 2.5 | 2.5 | 2.8 | 3.0 | 3.5 | 4.0 |
Drag the appropriate items to their respective bins.
Ionic
polar covalent
non-polar covalent
| S-I | H-Br | H-P | S-O | Be-F | Be-Cl | Sc-O |
Read by 3 people
11 Nov 2021
Iris MandiaLv9
22 Jan 2021
Already have an account? Log in