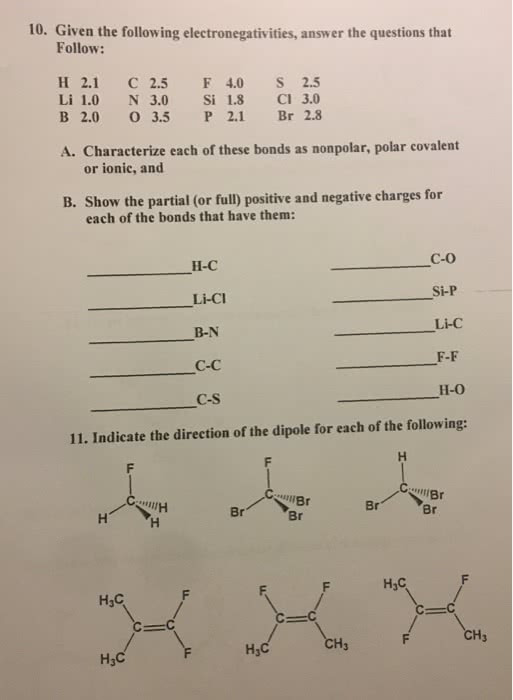
1
answer
0
watching
544
views
11 Nov 2019
A.Classify the bonds formed between each pair of elements based on electronegativity values in the following table:
Element Sc Be H P S I Br Cl O F Electronegativity 1.3 1.5 2.1 2.1 2.5 2.5 2.8 3.0 3.5 4.0
Drag the appropriate items to their respective bins.
Be and Cl, Be and F, S and I, S and O, H and Br,H and P, Sc and O
Ionic
Polar covalent
Nonpolar covalent
B.
Which of the following compounds has the highest boiling point?
Br2 NaBr HBr BrF
C.
Are all these Lewis symbols for Cl correct? (Figure 1)
Are all these Lewis symbols for correct? 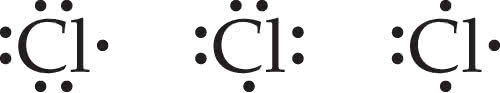
Yes, all symbols are correct. No, the first symbol is incorrect. No, the second symbol is incorrect. No, the third symbol is incorrect.
C CI: Cl-
A.Classify the bonds formed between each pair of elements based on electronegativity values in the following table:
| Element | Sc | Be | H | P | S | I | Br | Cl | O | F |
| Electronegativity | 1.3 | 1.5 | 2.1 | 2.1 | 2.5 | 2.5 | 2.8 | 3.0 | 3.5 | 4.0 |
Drag the appropriate items to their respective bins.
| Be and Cl, Be and F, S and I, S and O, H and Br,H and P, Sc and O Ionic Polar covalent Nonpolar covalent B. Which of the following compounds has the highest boiling point?
C. |
Are all these Lewis symbols for Cl correct? (Figure 1)
Are all these Lewis symbols for correct? 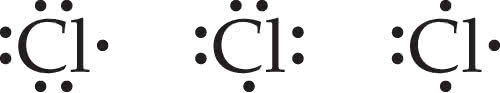
| Yes, all symbols are correct. |
| No, the first symbol is incorrect. |
| No, the second symbol is incorrect. |
| No, the third symbol is incorrect. |
C CI: Cl-
Jarrod RobelLv2
31 Oct 2019