0
answers
0
watching
196
views
12 Dec 2019
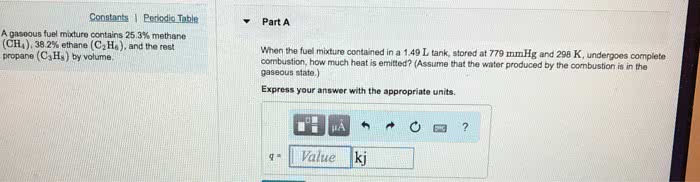
A gaseous fuel mixture contains 22.2% methane (CH4), 39.8% ethane (C2H6) and the rest propane (C3H8) by volume. When the fuel mixture contained in a 1.51 L tank, stored at 746 mmHg and 298 K, undergoes complete combustion, how much heat is emitted? (Assume that the water produced by the combustion is in the gaseous state.)
A gaseous fuel mixture contains 22.2% methane (CH4), 39.8% ethane (C2H6) and the rest propane (C3H8) by volume. When the fuel mixture contained in a 1.51 L tank, stored at 746 mmHg and 298 K, undergoes complete combustion, how much heat is emitted? (Assume that the water produced by the combustion is in the gaseous state.)