1
answer
0
watching
623
views
11 Nov 2019
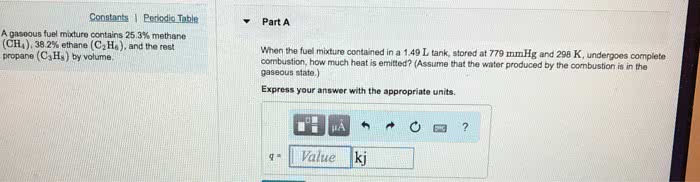
A gaseous fuel mixture stored at 745 mmHg and 298 K contains only methane(CH4) and propane (C3H8). When 12.8 L of this fuel mixture burns, it produces 775 kJ of heat. The enthalpy of combustion for CH4(g) is â802.5kJ. The enthalpy of combustion for C3H8(g) is â2043.9kJâ
What is the mole fraction of methane in the mixture? (Assume that the water produced by the combustion is in the gaseous state.)â
A gaseous fuel mixture stored at 745 mmHg and 298 K contains only methane(CH4) and propane (C3H8). When 12.8 L of this fuel mixture burns, it produces 775 kJ of heat. The enthalpy of combustion for CH4(g) is â802.5kJ. The enthalpy of combustion for C3H8(g) is â2043.9kJâ
What is the mole fraction of methane in the mixture? (Assume that the water produced by the combustion is in the gaseous state.)â
Patrina SchowalterLv2
4 Nov 2019