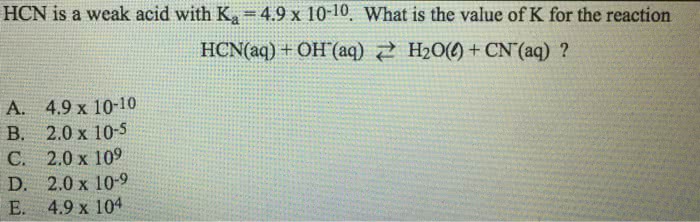1
answer
2
watching
829
views
taupegnu357Lv1
11 Dec 2019
Given the following acids and Ka values:
HClO4
HOAc
HCN
HF
1 x 107
1.76 x 10-5
4.93 x 10-10
3.53 x 10-4
What is the order of increasing base strength?
a. CN-, ClO4-, F-, OAc-
b. ClO4-, OAc-, CN-, F-
c. CN-, F-, OAc-, ClO4-,
d. ClO4-, F-, OAc-, CN-
e. CN-,OAc-, F-, ClO4-
Given the following acids and Ka values:
| HClO4 | HOAc | HCN | HF |
| 1 x 107 | 1.76 x 10-5 | 4.93 x 10-10 | 3.53 x 10-4 |
What is the order of increasing base strength?
a. CN-, ClO4-, F-, OAc-
b. ClO4-, OAc-, CN-, F-
c. CN-, F-, OAc-, ClO4-,
d. ClO4-, F-, OAc-, CN-
e. CN-,OAc-, F-, ClO4-
Greg MontoyaLv10
1 Dec 2020