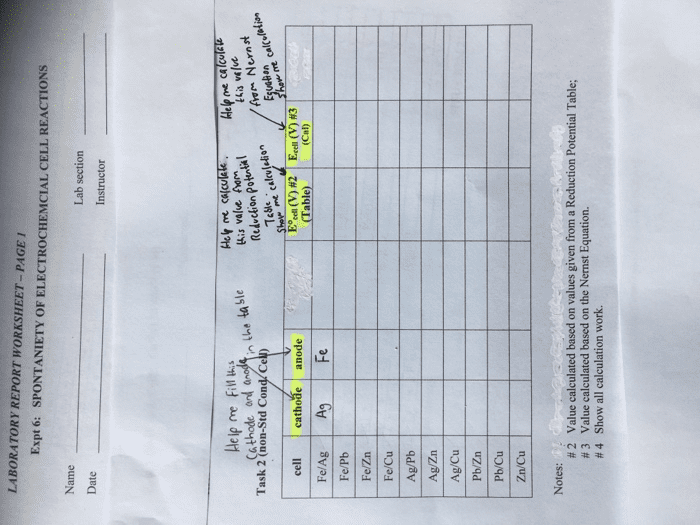
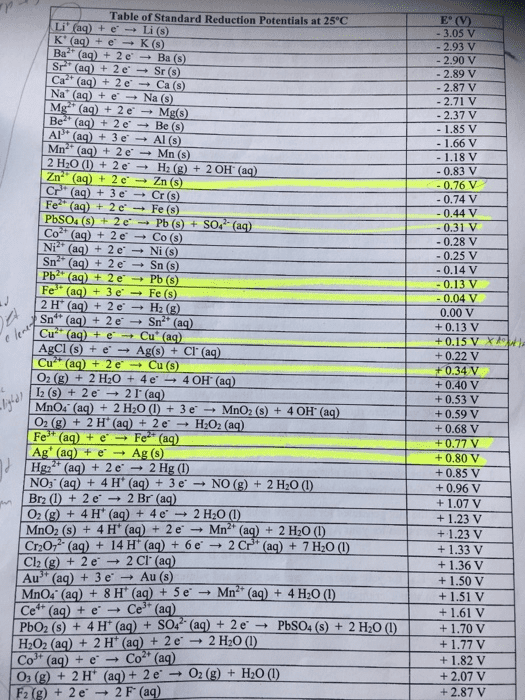
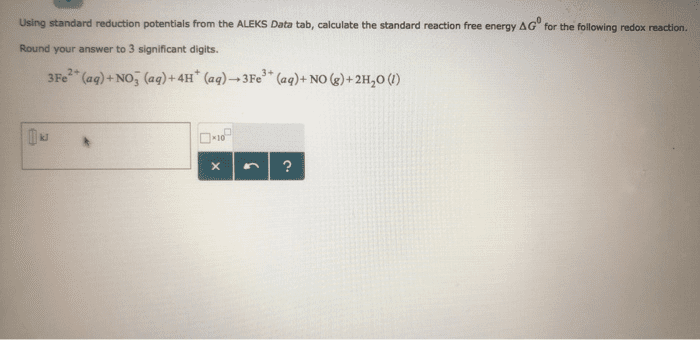
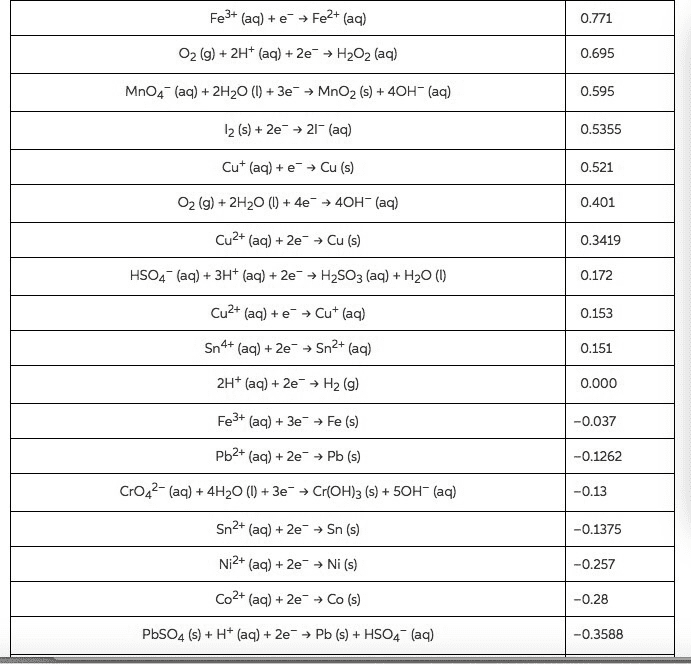
Compare table 1 (the standard reduction potentials) to the periodic trends (radius, Ionization energy, electron affinity, electronegativity). Do the potentials in the table agree with our periodic trends? Why/why not (use at least two examples in your argument)?
Table 1: Standard Reduction Potentials
Half-Reaction
E°, volts
Na+(aq) + eâ â Na(s)
-2.71
Al3+(aq) + 3 eâ â Al(s)
-1.66
Zn2+(aq) + 2 eâ â Zn(s)
-0.76
Cr3+(aq) + 3 eâ â Cr(s)
-0.74
Fe2+(aq) + 2 eâ â Fe(s)
-0.41
PbSO4(s) + 2 eâ â Pb(s) + SO42â(aq)
-0.36
Ni2+(aq) + 2 eâ â Ni(s)
-0.23
Sn2+(aq) + 2 eâ â Sn(s)
-0.14
Pb2+(aq) + 2 eâ â Pb(s)
-0.13
2 H+(aq) + 2 eâ â H2(g)
0
Sn4+(aq) + 2 eâ â Sn2+(aq)
0.15
AgCl(s) + eâ â Ag(s) + Clâ(aq)
0.22
Cu2+(aq) + 2 eâ â Cu(s)
0.34
O2(g) + 2 H2O(l) + 4 eâ â 4 OHâ(aq)
0.4
H2SO3(aq) + 4 H+(aq) + 4 eâ â S(s) + 3H2O(l)
0.45
Ag+(aq) + eâ â Ag(s)
0.8
O2(g) + 4 H+(aq) + 4 eâ â 2 H2O(l)
1.23
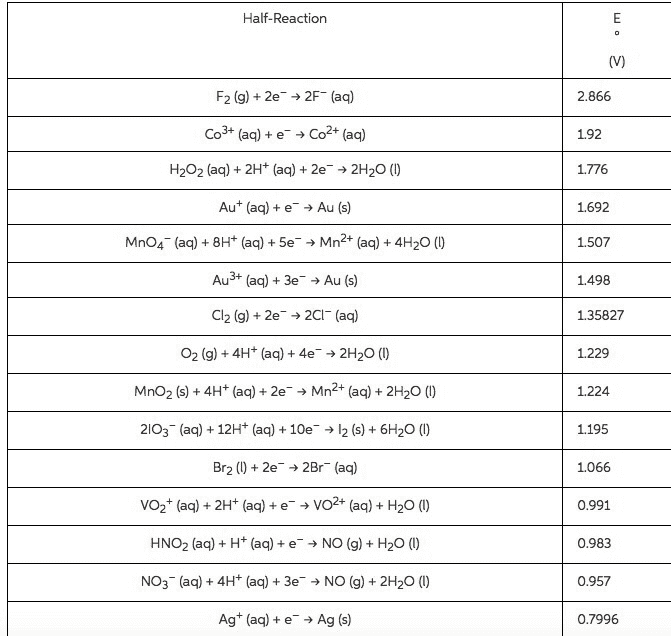
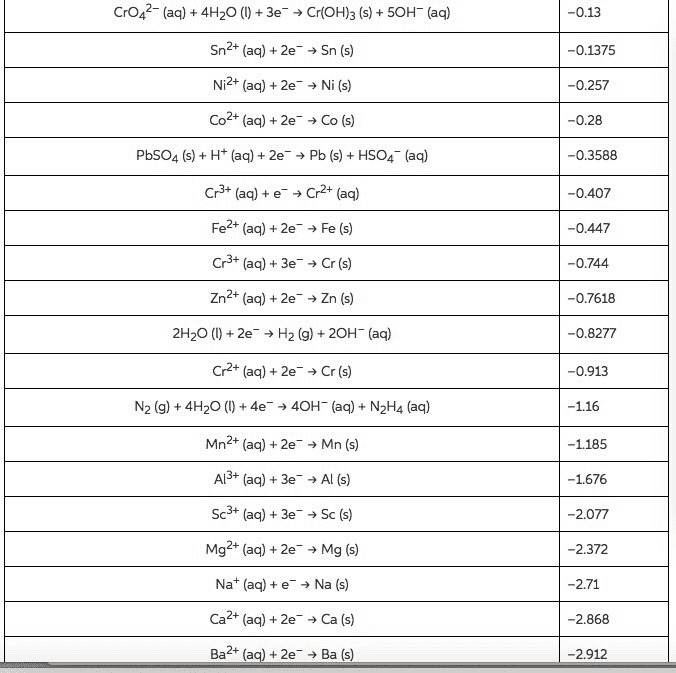
Compare table 1 (the standard reduction potentials) to the periodic trends (radius, Ionization energy, electron affinity, electronegativity). Do the potentials in the table agree with our periodic trends? Why/why not (use at least two examples in your argument)?
| Table 1: Standard Reduction Potentials | |
| Half-Reaction | E°, volts |
| Na+(aq) + eâ â Na(s) | -2.71 |
| Al3+(aq) + 3 eâ â Al(s) | -1.66 |
| Zn2+(aq) + 2 eâ â Zn(s) | -0.76 |
| Cr3+(aq) + 3 eâ â Cr(s) | -0.74 |
| Fe2+(aq) + 2 eâ â Fe(s) | -0.41 |
| PbSO4(s) + 2 eâ â Pb(s) + SO42â(aq) | -0.36 |
| Ni2+(aq) + 2 eâ â Ni(s) | -0.23 |
| Sn2+(aq) + 2 eâ â Sn(s) | -0.14 |
| Pb2+(aq) + 2 eâ â Pb(s) | -0.13 |
| 2 H+(aq) + 2 eâ â H2(g) | 0 |
| Sn4+(aq) + 2 eâ â Sn2+(aq) | 0.15 |
| AgCl(s) + eâ â Ag(s) + Clâ(aq) | 0.22 |
| Cu2+(aq) + 2 eâ â Cu(s) | 0.34 |
| O2(g) + 2 H2O(l) + 4 eâ â 4 OHâ(aq) | 0.4 |
| H2SO3(aq) + 4 H+(aq) + 4 eâ â S(s) + 3H2O(l) | 0.45 |
| Ag+(aq) + eâ â Ag(s) | 0.8 |
| O2(g) + 4 H+(aq) + 4 eâ â 2 H2O(l) | 1.23 |