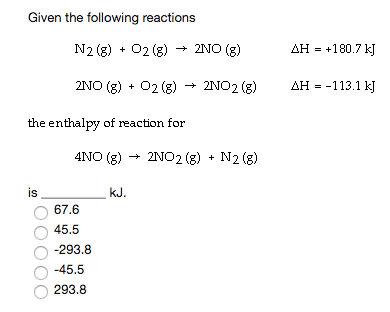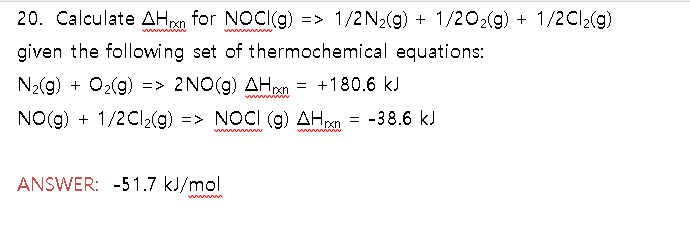1
answer
0
watching
135
views
18 Nov 2019
Find the molar enthalpy of formation of NO(g) from the following:

Find the molar enthalpy of formation of NO(g) from the following:


Find the molar enthalpy of formation of NO(g) from the following: N_2(g) + 2O_2(g) rightarrow 2NO_2(g) Delta H_rxn = 66.4 kJ 2NO(g) + O_2(g) rightarrow 2NO_2(g) Delta H_rxn = -114.1 kJ
Tod ThielLv2
31 Mar 2019