1
answer
0
watching
202
views
17 Nov 2019
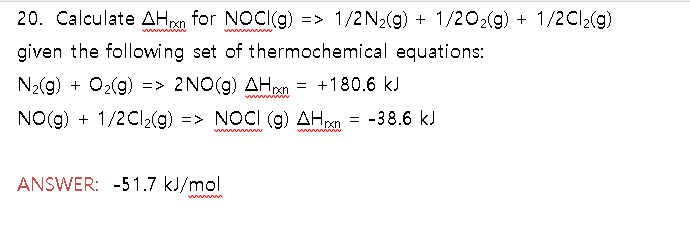
Calculate delta H_rxn for NOCl(g) 1/2 N_2(g) + 1/2 O_2(g) + 1/2 Cl_2(g) given the following set of thermochemical equations: N_2(g) + O_2(g) 2NO(g) delta H_rxn = +180.6 kJ NO(g) + 1/2 Cl_2(g) NOCI (g) delta H_rxn = -38.6 kJ
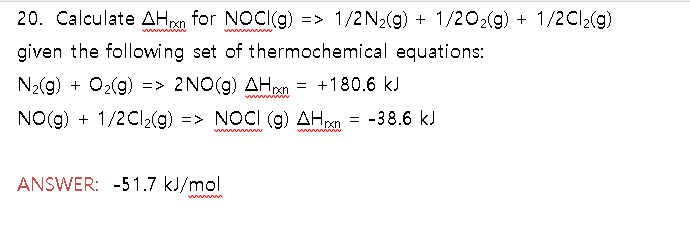
Calculate delta H_rxn for NOCl(g) 1/2 N_2(g) + 1/2 O_2(g) + 1/2 Cl_2(g) given the following set of thermochemical equations: N_2(g) + O_2(g) 2NO(g) delta H_rxn = +180.6 kJ NO(g) + 1/2 Cl_2(g) NOCI (g) delta H_rxn = -38.6 kJ
Irving HeathcoteLv2
17 Jan 2019
