1
answer
0
watching
474
views
18 Nov 2019
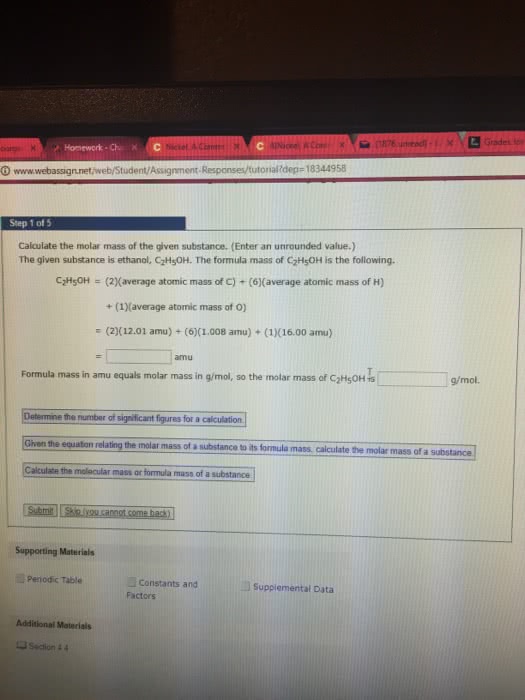
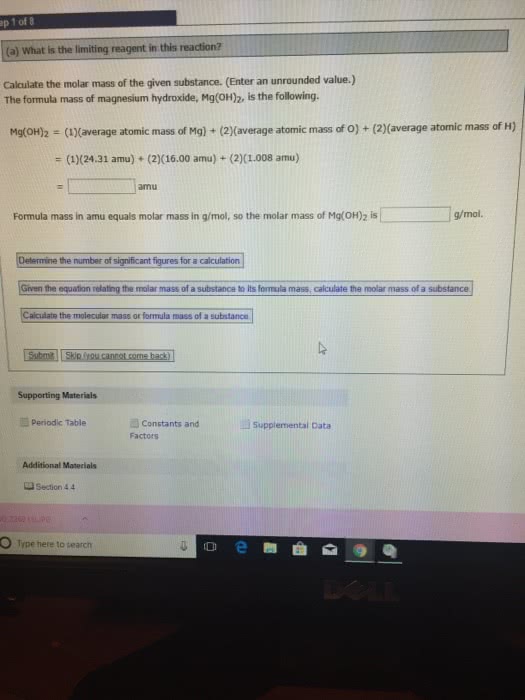
A sample of a substance with the empirical formula XBr2 weighs 0.5674 g. When it is dissolved in water and all its bromine is converted to insoluble AgBr by addition of an excess of silver nitrate, the mass of the resulting AgBr is found to be 0.9542 g. The chemical reaction is
XBr2 + 2 AgNO32AgBr + X(NO3)2
(a) Calculate the formula mass of XBr2.
Formula mass
XBr2 = g mol-1
(b) Calculate the atomic mass of X.
Atomic mass
X = g mol-1
A sample of a substance with the empirical formula XBr2 weighs 0.5674 g. When it is dissolved in water and all its bromine is converted to insoluble AgBr by addition of an excess of silver nitrate, the mass of the resulting AgBr is found to be 0.9542 g. The chemical reaction is
XBr2 + 2 AgNO32AgBr + X(NO3)2
(a) Calculate the formula mass of XBr2.
Formula mass
XBr2 = g mol-1
(b) Calculate the atomic mass of X.
Atomic mass
X = g mol-1
Nestor RutherfordLv2
23 Mar 2019