1
answer
0
watching
235
views
28 Sep 2019
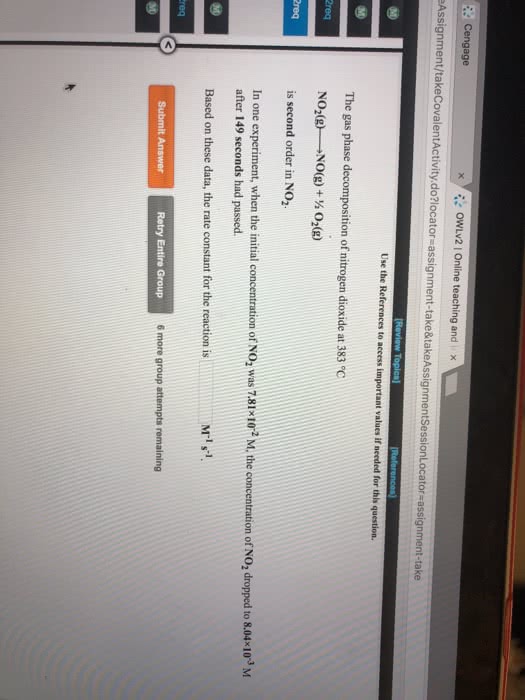
The gas-phase decomposition of NO2 was studied at 383oC resulting in the following data.a) Is the reaction first-order or second-order in NO2? Rxn: 2 NO2 2 NO + O2 Provide the rate expression in you answer.b) What is the value of the rate constant?Show me how you arrived at your conclustion.Time (s) [NO2], M0.0 0.10010.0 0.009020.0 0.0047
The gas-phase decomposition of NO2 was studied at 383oC resulting in the following data.a) Is the reaction first-order or second-order in NO2? Rxn: 2 NO2 2 NO + O2 Provide the rate expression in you answer.b) What is the value of the rate constant?Show me how you arrived at your conclustion.Time (s) [NO2], M0.0 0.10010.0 0.009020.0 0.0047
Patrina SchowalterLv2
28 Sep 2019