1
answer
0
watching
581
views
azurecrab717Lv1
9 Apr 2020
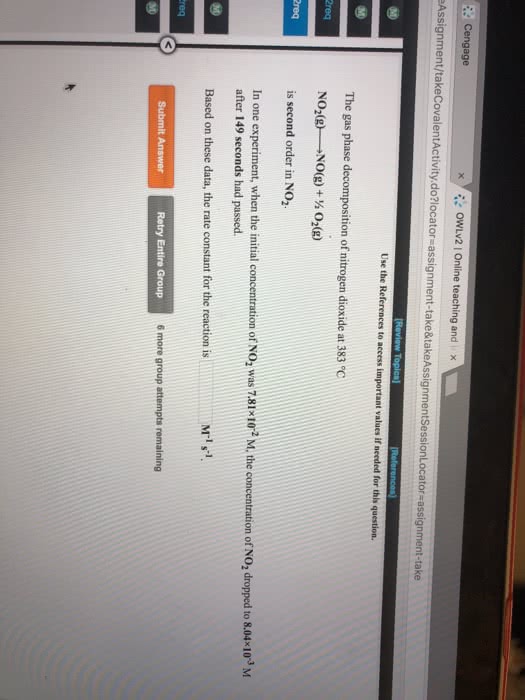
The gas-phase decomposition of NO2, 2 NO2(g)  2 NO(g) + O2(g), is studied at 383 °C, giving the following data:
2 NO(g) + O2(g), is studied at 383 °C, giving the following data:

(a) Is the reaction first order or second order with respect to the concentration of NO2? (b) What is the rate constant? (c) Predict the reaction rates at the beginning of the reaction for initial concentrations of 0.200 M, 0.100 M, and 0.050 M NO2.
The gas-phase decomposition of NO2, 2 NO2(g) 2 NO(g) + O2(g), is studied at 383 °C, giving the following data:
(a) Is the reaction first order or second order with respect to the concentration of NO2? (b) What is the rate constant? (c) Predict the reaction rates at the beginning of the reaction for initial concentrations of 0.200 M, 0.100 M, and 0.050 M NO2.
Sixta KovacekLv2
21 May 2020