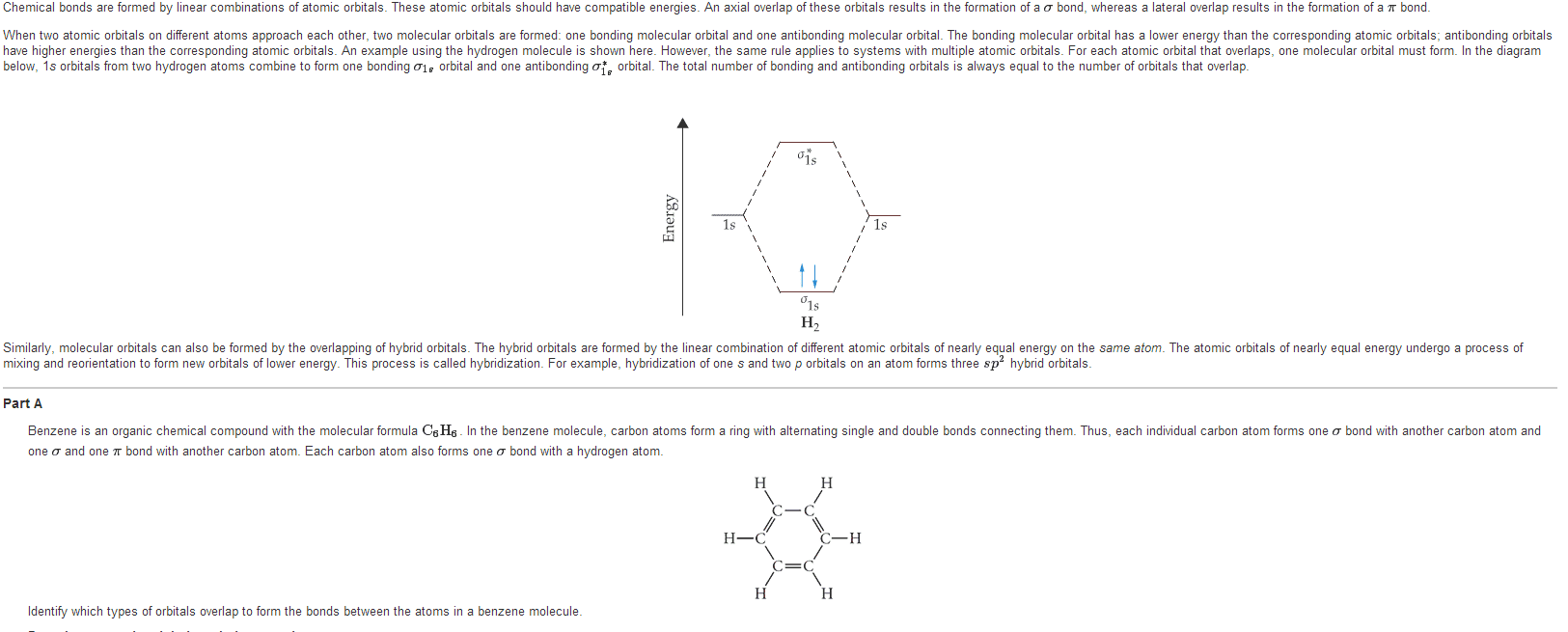
1
answer
0
watching
143
views
11 Nov 2019


(a) Explain with electron orbital configurations (2s and 2p) how the hybrid orbitals are formed in C_2H_2. Which orbital(s) are involved in forming the pi bond? (b) For the molecule shown below- (1) circle and identify all functional groups (except alkanes), (2) ID the hybridization of the atoms indicated with arrows (3) How many sigma bonds and pi bonds are present?
1
answer
0
watching
143
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Hubert KochLv2
21 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232